Not New. Spike --> Transferrin Receptor: B-cells, Tumors
Targeting Transferrin Receptor (mitochondrial dysfunction, cell death, immunosuppression) has a long history in the lab.
(Image: Diderot and d’Alembert Encyclopedia, 1751-1780)
-
Building on my other posts,
Not New:
In “gene therapies, researchers have begun to target polycations … with receptor ligands, like transferrin” (Hildebrandt et al. 2003, https://doi.org/10.1038/sj.gt.3301939)
“Amongst the numerous potential targets at the BBB, the transferrin receptor (TfR) remains the most common target used to ensure sufficient drug delivery to the brain” (Johnsen et al. 2019, doi: 10.1016/j.pneurobio.2019.101665).
“The most widely studied [receptor ligand for active targeting] is arguably the iron transferrin (TfR) receptor, as they are very highly expressed in the brain endothelium in comparison to the periphery, although the bone marrow, splenic and hepatocellular accumulation is always a concern (Lynch and Gobbo 2021, doi: 10.3390/nano11102632, citing Johnsen et al. 2019).
“Transferrin Receptor is another receptor for SARS-CoV-2 entry” (Tang X et al., held in preprint since 2020, https://doi.org/10.1101/2020.10.23.350348)
“The role of transferrin receptor 1 (TfR1) in SARS-CoV-2 infection has received little attention;” “Ferristatin II Efficiently Inhibits SARS-CoV-2 Replication in Vero Cells” (Sokolov et al. 2022, DOI: 10.3390/v14020317)
Kucia, M., Ratajczak, J., Bujko, K. et al. “Spike protein damages hematopoietic stem/progenitor cells” (Kuci et al. 2021, doi.org/10.1038/s41375-021-01332-z; article brought to attention by WMC Research Substack).
Here we will see:
Since 1993, cells (here, cancer, including B-cells) have been targeted for destruction using ligands for Transferrin Receptor (Elliott and Head 2012, http://dx.doi.org/10.4236/jct.2012.34039)
… for instance resulting in decreased Superoxide Dismutase (SOD) —> ROS, apoptosis (Elliott and Head 2012, citing Elliott et al. 1993, https://doi.org/10.1111/j.1749-6632.1993.tb17204.x)
“Reexposure to CoV [Spike] could induce reactive oxygen species (ROS) and decrease superoxide dismutase (SOD) in [antibody-producing plasma cells]”, which “showed apoptotic activities” and “antibody production declined quickly” (Mo et al. 2022, doi: 10.1155/2022/5397733).
“The regulation of antibody production is linked to the generation and maintenance of plasmablasts and plasma cells from their B cell precursors” (Nutt et al. 2015, doi: 10.1038/nri3795)
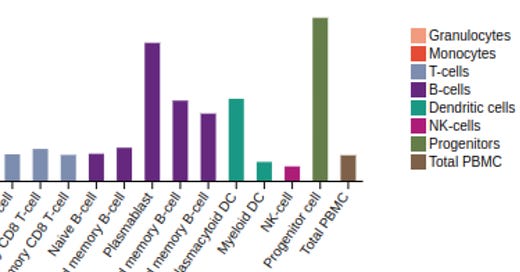
Highest expression of Transferrin Receptor 1 is in bone marrow progenitor and B cells (Human Protein Atlas 2022).
Too large to cover in this post:
Tumor cells highly express Transferrin Receptor (described by Elliott and Head 2012).
When Spike enters cells, it enters the nucleus and has been shown to impair VDJ recombination and reduce p53 (Jiang H and Mei YF 2021, https://doi.org/10.3390/v13102056, recently retracted without substantiation, reviewed by Arkmedic substack '“Welcome to Gilead”.)
Kanduk (2021, doi: 10.1055/s-0041-1735590) shows “an impressively high level of molecular mimicry exists between SARS-CoV-2 spike gp and tumor-associated proteins. Numerically, 294 tumor-suppressor proteins share 308 pentapeptides with the viral antigen… which might equate to induce or enhance carcinogenesis in almost all of the human organs” (see coverage by DoorlessCarp substack, “Autoimmune disorders: COVID-19, spike protein & homologous epitope” / “Tumor Suppressor Protein p53 & BRCA related cancers”).
-
Cancer: Tumor Iron Metabolism, Mitochondrial Dysfunction and Tumor Immunosuppression; “A Tight Partnership—Was Warburg Correct?”
Elliott RL and Head JF. Journal of Cancer Therapy, 2012, 3, 278-311. http://dx.doi.org/10.4236/jct.2012.34039
“… After nearly 40 years of research and treating breast cancer patients, we have become convinced that the three major players in tumorigenesis are: 1) Tumor iron metabolism; 2) Mitochondrial dysfunction; and 3) Tumor immune suppression. … We suspect that an initial event in tumorigenesis may be some slight dysfunction in iron metabolism…
Tumor Iron Metabolism
Abnormal iron metabolism may be the main culprit and player in tumorigenesis, as it is definitely involved in tumor metabolism and cancer mitochondrial dysfunction. Iron also plays a significant role in tumor immunosuppression…. Transferrin (TF) … transports iron [7]. Iron bound TF specifically interacts with a cell surface transferrin receptor (TFR1) that promotes the transport of iron across the cell membrane. …Lazarus and Baines [12] documented that transferrin receptors were present on malignant and proliferating cells in high numbers …
In 1993, we [16] reported on the role of iron metabolism in breast carcinoma. … This study … confirmed that transferrin could be used as a carrier to target toxic therapy selectively to tumor tissue. There was strong evidence that our platinum transferrin complex (MPTC-63) decreases metallothionein synthesis, resulting in a decrease of superoxide dismutase activity. The decrease in superoxide dismutase activity within the cell results in increased susceptibility of the cell to superoxide anions and other free radicals. …
The Penichet group from UCLA have done a great job in reporting on the role of the TFR in cancer [21,22]. They discuss the elevated levels of the TFR in malignancies, its relevance in cancer, and that the extracellular accessibility of this molecule make it an excellent antigen for the treatment of cancer using antibodies [21]. They show that the TFR can be targeted by monoclonal antibodies specific for the extracellular domain of the receptor. These cytotoxic antibodies targeting the TFR inhibit cell growth, and also induce apoptosis in the targeted malignant cells [21]. In Part II of their work on targeting the TFR, they discuss in detail targeted delivery of many different therapeutic agents into cancer cells [22]. … Designing targeted therapies …can be done by binding or conjugating cytotoxic agents to TF or directing antibodies and antibody complexes to the TFR. Daniels and Ortiz-Sanchez et al. [23] of the Penichet group have shown the tremendous importance of the TFR in B-cell malignancies. They showed in vitro and in vivo efficacy of an antibody multifaceted approach targeting the human TFR for the treatment of B-cell malignancies.”
-
-
1993: Targeting Transferrin Receptor with something toxic —> Decreased Superoxide Dismutase (with a goal of cell death)
-
Above: Targeting something toxic (platinum transferrin complex MPTC-63) to Transferrin Receptor resulted “in a decrease of superoxide dismutase activity. The decrease in superoxide dismutase activity within the cell results in increased susceptibility of the cell to superoxide anions and other free radicals” (Elliott and Head 2012, citing Elliott et al. 1993, https://doi.org/10.1111/j.1749-6632.1993.tb17204.x).
Question: Was this reduction in superoxide dismutase specific to platinum / MPTC-63, or could that be repeated with other toxic / signaling / immunogenic cargo?
Below: Exposure to Spike protein suppresses superoxide dismutase expression in antibody-producing plasma cells (Mo et al. 2022 doi: 10.1155/2022/5397733).
-
-
2022: Superoxide Dismutase Prevents SARS-CoV-2-Induced Plasma Cell Apoptosis and Stabilizes Specific Antibody Induction.
Mo LH, Luo XQ, Ma K, Shao JB, Zhang GH, Liu ZG, Liu DB, Zhang HP, Yang PC. Oxid Med Cell Longev. 2022 Jan 17;2022:5397733. doi: 10.1155/2022/5397733.
[These authors say “SARS-CoV-2 viral spike protein (CoV, in short)” instead of “Spike.” For clarity to readers and to maintain standardization with other published studies, I have inserted “[Spike]” following CoV.]
3.4. Exposure to CoV [Spike] Suppresses SOD Expression in sPCs [antibody-producing plasma cells] through the JAK2/STAT1 Pathway
“…Exposure to CoV [Spike] induces plasma cell apoptosis and suppresses SOD in plasma cells. As the Janus kinase (JAK)2/STAT1 … pathway activation is associated with cell apoptosis initiation [9], the JAK2/STAT1 activation in sPCs was assessed after exposing to CoV [Spike] overnight. The results showed that CoV [Spike] exposure markedly induced the JAK2 and STAT1 phosphorylation in sPCs …
Abstract: “… Currently, vaccines against COVID-19 have been employed in the general population to prevent COVID-19 infection. The efficacy of this vaccination has to be verified in further investigation. … We induced the anti-CoV [Spike antibodies] Ab in mice. However, we observed that the Ab production declined quickly in mice after the immunization. Further experimental evidence showed apoptotic activities in the [Spike] CoV-specific *Ab-producing plasma cells* (sPCs). Therefore, we carried out this project. The results showed that reexposure to CoV [Spike] could induce reactive oxygen species (ROS) and decrease superoxide dismutase (SOD) in sPCs.”
[ROS + decreased superoxide dismutase —> cancerous changes, cell death (“apoptotic activities”) …]
Antibody-Producing Plasma Cells / B Cells:
Above, Mo et al. (2022) found ROS increase, Superoxide Dismutase reduction, and Apoptosis in antibody-producing plasma cells following repeated Spike reexposure.
Above, UCLA Penichet group targeted TFR Transferrin Receptor in order to kill B-cells / used cytotoxic monoclonal antibodies specific for the extracellular domain of the TFR receptor (Elliott and Head 2012)
Above, Tang et al. (2020) found that ‘Transferrin Receptor is another receptor for SARS-CoV-2 entry’
Below, Antibody-producing plasma cells / plasmablasts / B cell precursors + Transferrin Receptor expression. Transferrin Receptor is a target of Spike.
-
The generation of antibody-secreting plasma cells
Nutt SL, Hodgkin PD, Tarlinton DM, Corcoran LM. Nat Rev Immunol. 2015 Mar;15(3):160-71. doi: 10.1038/nri3795.
“The regulation of antibody production is linked to the generation and maintenance of plasmablasts and plasma cells from their B cell precursors…”
-
-
Precursors / Progenitors / Plasmablasts:
Expression of Transferrin Receptor 1 (The Human Protein Atlas)
Image: TFRC Transferrin Receptor 1 expression in immune cells, detail view (The Human Protein Atlas, July 2022). Progenitor cells and B-cells, including Plasmablasts, show highest expression.
Image: TFRC Transferrin Receptor 1 expression in immune cells (The Human Protein Atlas, July 2022). See enlarged detail above.
-
Image: TFRC Transferrin Receptor 1 expression in various tissues (The Human Protein Atlas, July 2022). The tallest gray bar is bone marrow.
-
-
What might happen if Spike enters these cells?
This article was brought to attention in a post by WMC Research, “DOES THE SPIKE PROTEIN ABROGATE CENTRAL TOLERANCE IN HIGH-AVIDITY AUTOREACTIVE B-CELLS VIA PI3K ACTIVATION”.
Articles cited here are for non-profit education and research only; please refer to the copyrights of their owners.







Ferroptosis related pathophysiology hasn't gone away, it's not discussed enough thanks, multiple oxidative pathways at work.
Ferroptosis: mechanisms and links with diseases
https://www.nature.com/articles/s41392-020-00428-9
Thank you both for the comments.
I'm turning them off for the moment because the article just reached a pleasantly precise PDF-printable length.