Not New. Arginine / HIV-TAT lab tool --> Syncytium-building, immunodeficiency, sudden death.
Follow the Science. Spike Arginine: TAT-ACE2, Cell Penetrating Peptides, Furin cleavage, gene delivery, BBB, tumor-targeting, lymphopenia, VAIDS, syncytia, giant clots, fusion inhibitors, brain stem
Spike features arginine repeats. This article’s (arguable lack of) focus is historically-HIV1-Tat-inspired multi-Arginine sequences - their long history in the lab for e.g. gene delivery and their potential pathological effects including syncytia-building. This will be connected to their known neuropathological effects and to Spike presence in the brainstem, questioning whether disruption [electrical conductance, syncytia-building, extracellular matrix derangement] contributes to unexplained clot masses and / or sudden death.
Update Dec. 2022: Linked at the bottom of the page are posts by Dr. McCullough and James Lyons-Weiler now mentioning this theory I put out about Spike causing the giant clots via Syncytia-building. Also: intriguing post by Charles Wright describing patents held by Malone for cationic (e.g. Arginine) transfection and a link with HIV-vaccine attempts.
-
Bat soup from the market
Image: Woodcut (1834) by John Ashton (urn:oclc:record:1041814191). Thanks to Jon Crabb for highlighting it in his article, https://publicdomainreview.org/essay/woodcuts-and-witches
Rough Overview:
HIV-1 TAT (featuring arginine motifs)
TAT, ACE2
Arginine-rich motifs (cell-penetrating peptides / nano, DNA, RNA delivery / Furin cleavage site)
Syncytia-building
[Lymphocyte elimination] / [Acquired Immunodeficiency]
Trying to block Syncytia
[Brain pathologies following TAT, Spike] / [Spike in Brainstem]
[Sudden death]
Next post: Expanding on Arginine / Tat brain and tissue pathologies with a look at Extracellular Matrix disbalance.
-
-
HIV-1 TAT (featuring Arginine)
Let’s start at a familiar topic to my Substack: lab tools designed to penetrate the protective BBB [otherwise known as ‘stays in the arm’].
-
Not New. Nano-micelles, PEG, RNA, HIV motifs, tumors, intranasal, brain….
2013: Delivery of siRNA to the brain using a combination of nose-to-brain delivery and cell-penetrating peptide-modified nano-micelles
T. Kanazawa, F. Akiyama, S. Kakizaki, Y. Takashima, Y. Seta. 2013. https://doi.org/10.1016/j.biomaterials.2013.08.036
… “To improve the efficiency of small interfering RNA (siRNA) delivery to the brain, we developed a nose-to-brain delivery system combined with cell-penetrating peptide (CPP) modified nano-micelles comprising polyethylene glycol–polycaprolactone (PEG–PCL) copolymers conjugated with the CPP, Tat (MPEG–PCL–Tat). … Intranasal delivery of dextran with MPEG–PCL–Tat improved brain delivery compared to intravenous delivery of dextran either with or without MPEG–PCL–Tat. We also studied the intranasal transfer of MPEG–PCL–Tat to the brain via the olfactory and trigeminal nerves, the putative pathways to the brain from the nasal cavity. We found that MPEG–PCL–Tat accelerated transport along the olfactory and trigeminal nerve pathway because of its high permeation across the nasal mucosa…
The intracellular uptake of macromolecules, such as proteins and genes, can be enhanced by cell-penetrating peptides such as the HIV-1 Tat peptide [14] ... The Tat peptide is rich in arginine and has 6 arginine and 2 lysine residues in a 13-amino-acid stretch. …
In a previous study, we demonstrated that Tat, a cell-penetrating peptide derived from HIV-Tat, and the modified amphiphilic block copolymers of poly (ethylene glycol) and poly (ε-caprolactone) (MPEGePCLeTat) are capable of forming stable complexes with pDNA and siRNA. Furthermore, MPEGePCLeTat efficiently delivers pDNA and siRNA into tumor tissues and exerts significantly higher gene expression or gene silencing in tumor tissues following systemic delivery [22,23]…
Diagram: T. Kanazawa et al. / Biomaterials 34 (2013) 9220e9226
… The dynamics of distribution within the olfactory bulb, rostral brain tissue, caudal brain tissue, and brainstem after intranasal administration of MPEGePCLeTat/Alexa-dextran indicated that the rostral brain tissue had a high distribution, which appeared to follow a distribution trend into the olfactory bulb while the caudal brain tissue and brainstem had high distribution that appeared to arise as a result of transport via the trigeminal nerve. This suggested that, once transported to the olfactory bulb and brainstem via the olfactory nerve and trigeminal nerve pathways, nucleic acid is also transferred to other brain tissues.”
-
2012: Molecular mechanisms in the dramatic enhancement of HIV-1 Tat transduction by cationic liposomes.
Li GH, Li W, Mumper RJ, Nath A.FASEB J. 2012 Jul;26(7):2824-34. doi: 10.1096/fj.11-203315.
-
2009: Immobilized HIV-1 Tat protein promotes gene transfer via a transactivation-independent mechanism which requires binding of Tat to viral particles.
Nappi F, Chiozzini C, Bordignon V, Borsetti A, Bellino S, Cippitelli M, Barillari G, Caputo A, Tyagi M, Giacca M, Ensoli B.J Gene Med. 2009 Nov;11(11):955-65. doi: 10.1002/jgm.1381.
-
Nuclear accumulation… Arginine [R] motifs.
2004: Furin cleavage of the HIV-1 Tat protein.
Tikhonov I, Ruckwardt TJ, Berg S, Hatfield GS, David Pauza C. FEBS Lett. 2004 May 7;565(1-3):89-92. doi: 10.1016/j.febslet.2004.03.079.
“Extracellular human immunodeficiency virus-1 (HIV-1) Tat protein and Tat-derived peptides are biologically active … We identified two putative furin cleavage sites and showed that Tat protein was cleaved in vitro at the second site, RQRR … Furin processing did not affect the rates for Tat uptake and *nuclear accumulation* in HeLa or Jurkat cells …”
-
*Nuclear accumulation is key to why Tat and its Arginine-rich derivatives have been so frequently in lab experiments for gene delivery etc. Visualized here in Astrocytes (which will be important in the last sections of this post)
The taming of the cell penetrating domain of the HIV Tat: myths and realities
Chauhan A, Tikoo A, Kapur AK, Singh M. Review. J Control Release. 2007 Feb 12;117(2):148-62. DOI: 10.1016/j.jconrel.2006.10.031
“Fig 2. Nuclear expression of Tat in SVGA (astrocytic) cells. … (A) Nuclear localization of Tat in two daughter cells. (B) DAPI nuclear staining corresponds to cells in panel (A).
…PTD [protein transduction domain] is responsible for nuclear retention of Tat protein due to the presence of strong *nuclear localization signal* …and hence will direct the fusion protein to the nucleus…”
-
*Nuclear localization signal is also in Spike: see small print in red. Notice the RR Arginine-rich Tat-offspring sequence
Nuclear translocation of spike mRNA and protein is a novel pathogenic feature of SARS-CoV-2
Sarah Sattar, Juraj Kabat, Kailey Jerome, Friederike Feldmann, Kristina Bailey, Masfique Mehedi. bioRxiv 2022.09.27.509633; doi: https://doi.org/10.1101/2022.09.27.509633
-
*Penetrating the nucleus: Spike in nucleus was first shown by Jiang and Mei. Its improper retraction is analyzed on ArkMedic’s Substack, which also highlighted the above article by Sattar et al.
SARS-CoV-2 Spike Impairs DNA Damage Repair and Inhibits V(D)J Recombination In Vitro.
Jiang, H.; Mei, Y.-F. Viruses 2021, 13, 2056 [retraction of: Viruses. 2021 Oct 13;13(10):]. Viruses. 2022;14(5):1011. doi:10.3390/v14051011
Image Jiang and Mei 2021: blue = nucleus, green = Spike protein.
“We report that the SARS–CoV–2 spike protein significantly inhibits DNA damage repair, which is required for effective V(D)J recombination in adaptive immunity. Mechanistically, we found that the spike protein localizes in the nucleus and inhibits DNA damage repair by impeding key DNA repair protein BRCA1 and 53BP1 recruitment to the damage site. Our findings … underscore the *potential side effects of full-length spike-based vaccines*.
*Oops — Retracted
-
-
Not New. Arginine motifs, TAT, PEG…
2009: TAT peptide and its conjugates: proteolytic stability
Jacob Grunwald, Tomas Rejtar, Rupa Sawant, Zhouxi Wang, Vladimir P Torchilin. Bioconjug Chem. 2009 Aug 19;20(8):1531-7. DOI: 10.1021/bc900081e
The proteolytic cleavage of TATp, TATp-PEG(1000)-PE conjugate (TATp-conjugate), and TATp as TATp-conjugate in mixed micelles made of TATp-conjugate and PEG(5000)-PE … were studied …. to simulate the proteolytic activity of human plasma. …The main initial fragmentation is an endocleavage at the carboxyl terminus resulting in an Arg-Arg (RR) dimer. …TATp in TATp-Mic was highly protected against proteolysis with an over 100-fold increase in half-life .... The shielding of TATp by PEG moieties in the proposed TATp-Mic is of great importance for its potential use as a cell-penetrating moiety for multifunctional "smart" drug delivery systems with detachable PEG.
-
Reminder: PEG in current delivery systems:
Assessment report COVID-19 Vaccine Moderna, Common name: COVID-19 mRNA Vaccine (nucleoside-modified).
European Medicines Agency, Committee for Medicinal Products for Human Use. 11 March 2021 EMA/15689/2021 Corr.1*1. Procedure No. EMEA/H/C/005791/0000
“The finished product … contains an mRNA active substance … that encodes for the pre-fusion stabilised spike glycoprotein of 2019-novel Coronavirus (SARS-CoV-2) encapsulated into lipid nanoparticles (LNP) …. The LNP are composed of four lipids which act as protectants and carriers of the mRNA. These are: heptadecan-9-yl 8-((2-hydroxyethyl)(6-oxo-6-(undecyloxy)hexyl)amino)octanoate (SM-102, a custommanufactured, ionisable lipid), 1,2-dimyristoyl-rac-glycero-3-methoxypolyethylene glycol-2000 (PEG2000-DMG), 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) and cholesterol.
PEG2000 DMG… is a novel excipient, not previously used in an approved finished product within EU.”
-
-
Suggested HIV-1 Tat targeting of ACE2:
Lipid-based nano delivery of Tat-peptide conjugated drug or vaccine-promising therapeutic strategy for SARS-CoV-2 treatment
Ansari MA et al. 2020. doi: 10.1080/17425247.2020.1813712
Image shows nanoformulation-based Tat-peptide targeting ACE2 or Spike. What else is a ligand for ACE2?
Cell-penetrating peptides (CPPs) based-nano delivery of repurposed drugs
“Most importantly, the nano delivery of repurposed antiviral drugs can be further enhanced by conjugating to cell-penetrating peptides (CPPs). The discovery of CPPs, a short cationic peptide with a high content of basic amino acid residues [10], readily facilitate intracellular intake and delivery of a variety of nanosize, small and large molecules into cytoplasm or nucleus [11,12]… The ‘cargo’ of these molecules is either due to electrostatic forces between the positively charged TAT-peptide and negative charges of phospholipids membrane** or nonelectrostatic hydrogen or hydrophobic interactions [12,13]…”
** Think: antiphospholipid antibodies arising as a result of spike exposure + hypercoaguability of antiphospholipid syndrome / antibodies: if an antigen is designed to attach to the phospholipids, they are in the scope of defensive antibody building **…
“… CPPs deliver the cargo into the cell either through macropinocytosis, caveolae-mediated endocytosis or clathrin-independent endocytosis mechanism. It has also been studied that HIV1 TAT-peptides directly penetrate the membranes by generating nanoscale pores [13]. CPPs has several advantages over other delivery and translocation approaches as it is inexpensive, easy to manufacture and usually nontoxic [14]. They have a higher capability to translocate into a wide range of cell types, higher rate of cellular permeability and uptake, more comfortable to pass *other biological barriers*… [14,15].”
*See my entries on bypassing the blood brain barrier - also relevant to reproductive organs.
“… It has been reported that CPP-based drug delivery systems have reached clinical trials for cancer diagnosis and therapy, where they showed enhanced efficacy [18]. It is, therefore, reasonable to expect similar progress in the development of nanoformulation-based CPP-conjugated nano delivery of repurposed antiviral drugs for the treatment of COVID-19. The HIV-1 TAT-peptide47-57 (GRKKRRQRRRP)*, a short cationic (8 positive charges) and high content of essential amino acid (2 lysine and 6 arginine), rapidly became a popular and powerful research tool to enhance the transport and delivery of proteins, DNA/RNA, viruses, drugs and nanoparticles inside the cells [12,15]."
*R = the abbreviation for Arginine
“…According to Milken Institute, ~123 vaccines, ~21 repurposed antiviral drugs, ~58 antibodies, ~15 cell-based, 6 RNA-based and more than 80 such as immune enhancers, immune-modulating, antimalarial, antiparasitic and anti-inflammatory are at different stages of clinical trials for the treatment of COVID-19 infections [19]. Unluckily, none of the ongoing clinical trials and treatments is based on nanocarriers and Tat-peptide conjugated-nanoformulation strategies despite the many advantages in nanocarriers and Tat-peptide drug delivery systems. Therefore, we suggest that the efficacy of antiviral activity of repurposed drug or vaccine against COVID-19 can be improved and enhanced by conjugating it to the Tat-peptides by utilizing nanoformulation-based nanocarriers delivery systems. We do not have high-level biosafety facilities for testing or analyzing the antiviral efficacy of nanoformulations-based nanocarriers for the delivery of Tat-peptide conjugated repurposed drug or vaccine using live SARS-CoV-19 either in vitro or in vivo. …
Select Citations:
10. Silva S, Almeida AJ, Vale N. Combination of cell-penetrating peptides with nanoparticles for therapeutic application: a review. Biomolecules. 2019;9:22. [PMC free article]
11. Zhao H, Wu M, Zhu L, et al. Cell-penetrating peptide-modified targeted drug-loaded phase-transformation lipid nanoparticles combined with low-intensity focused ultrasound for precision theranostics against hepatocellular carcinoma. Theranostics. 2018;8:1892. [PMC free article]
12. Quan X, Sun D, Zhou J. Molecular mechanism of HIV-1 TAT peptide and its conjugated gold nanoparticles translocating across lipid membranes. Phys Chem Chem Phys. 2019;21:10300–10310. [PubMed]
13. Ciobanasu C, Siebrasse JP, Kubitscheck U. Cell-penetrating HIV1 TAT peptides can generate pores in model membranes. Biophys J. 2010;99:153–162. [PMC free article]
14. Temsamani J, Vidal P. The use of cell-penetrating peptides for drug delivery. Drug Discov Today. 2004;9:1012–1019. [PubMed]
15. Skwarczynski M, Toth I. Cell-penetrating peptides in vaccine delivery: facts, challenges and perspectives. Ther Deliv. 2019;10:465–467. [PubMed]
-
-
ARGININE
HIV-1 Tat’s success seems to be based on Arginine. Decades of studies have been making small adjustments to the Tat sequence and observing comparable results: to what extent are the physiological effects of Tat in vivo comparable to an adjusted Arginine-rich sequence in vivo? Spike has Arginines [R] in strategic locations. Following this unresolved question, some studies on Tat’s in vivo effects are mentioned at the end of this post.
-
2003: Membrane permeability commonly shared among arginine-rich peptides.
Futaki S, Goto S, Sugiura Y.J Mol Recognit. 2003 Sep-Oct;16(5):260-4. doi: 10.1002/jmr.635.PMID: 14523938 Review.
Delivery of proteins and other macromolecules using membrane-permeable carrier peptides is a recently developed novel technology, which enables us to modulate cellular functions for biological studies with therapeutic potential. One of the most often used carrier peptides is the arginine-rich basic peptide derived from HIV-1 Tat protein [HIV-1 Tat (48-60)]. Using this peptide, efficient intracellular delivery of molecules including proteins, oligonucleic acids and liposomes has been achieved. We have demonstrated that these features were commonly shared among many arginine-rich peptides such as HIV-1 Rev (34-50) and octaarginine. Not only the linear peptides but also branched-chain peptides showed efficient internalization with an optimum number of arginines (approximately eight residues). The structural and mechanistic features of the translocation of these membrane-permeable arginine-rich peptides are reviewed.
-
-
Transferrin* and octaarginine modified dual-functional liposomes with improved cancer cell targeting* and enhanced intracellular delivery for the treatment of ovarian cancer.
Deshpande P, Jhaveri A, Pattni B, Biswas S, Torchilin V.Drug Deliv. 2018 Nov;25(1):517-532. doi: 10.1080/10717544.2018.1435747.
*See my other entries on Spike targeting Transferrin Receptor. If these tools indeed demonstrate improved tumor targeting, consider recent tumor statistics and Kanduc’s findings of pro-carcinogenic motifs hidden in Spike: (“From Anti-Severe Acute Respiratory Syndrome Coronavirus 2 Immune Response to Cancer Onset via Molecular Mimicry and Cross-Reactivity,” 2021. doi: 10.1055/s-0041-1735590), covered on DoorlessCarp’s Substack.
-
-
Speaking of Arginine… Furin cleavage site (surprise). R = arginine abbreviation.
Waves and variants of SARS-CoV-2: understanding the causes and effect of the COVID-19 catastrophe
Thakur, V., Bhola, S., Thakur, P. et al. Infection 50, 309–325 (2022). https://doi.org/10.1007/s15010-021-01734-2
“…The interplay between furin and spike glycoprotein is very fascinating, as furin plays a crucial role in SARS-CoV-2 entry and severity in host cells, and therefore reduces the need of other proteases for entry. The focus on the infectivity of SARS-CoV-2 with furin has been solely on the preactivation by furin (proprotein convertase). The protein cleavage site in spike glycoprotein of SARS-CoV-2 in S1/S2 site 1 is arginine (RR) multibasic, i.e. SPRRARSVAS at C terminal side …Surprisingly, the SARS-CoV-2 spike protein has a multibasic cleavage site containing arginine residues on the first and fourth position, which efficiently enhance proteolytic cleavage of S protein with ACE-2 receptor by furin due to the presence of extra arginine at the P4 position that enhances infection.
… The spike protein is cleaved by host type II transmembrane serine proteases (TMPRSS2) and furin at the S1/S2 polybasic cleavage site (PRRA) having arginine residues that empower the high cleavability (S2) [16, 17].”
-
-
Not New. Multi-basic multiple Arginine residues, HIV TAT, gene delivery…
2015: Cationic Cell-Penetrating Peptides Are Potent Furin Inhibitors
Ramos-Molina B, Lick AN, Nasrolahi Shirazi A, Oh D, Tiwari R, El-Sayed NS, Parang K, Lindberg I. PLoS One. 2015 Jun 25;10(6):e0130417. doi: 10.1371/journal.pone.0130417.
“Cationic cell-penetrating peptides have been widely used to enhance the intracellular delivery of various types of cargoes, such as drugs and proteins. These reagents are chemically similar to the multi-basic peptides that are known to be potent proprotein convertase inhibitors. Here, we report that both HIV-1 TAT47-57 peptide and the Chariot reagent are micromolar inhibitors of furin activity in vitro. … In addition, cyclic polyarginine peptides containing hydrophobic moieties which have been previously used as transfection reagents also exhibited potent furin inhibition in vitro and also inhibited intracellular convertases. Our finding that cationic cell-penetrating peptides exert potent effects on cellular convertase activity should be taken into account when biological effects are assessed…
Cationic peptides present within envelope proteins are used by many viruses to gain entry into host cells. These peptides, which efficiently pass through the plasma membrane and either remain in the cytoplasm or reach the nucleus, are frequently used as protein transduction reagents* (reviewed in [1,2])….
*Pausing here to highlight what protein transduction is:
Returning to Ramos-Molina et al. 2015:
… “One of the most studied CPPs [cell-penetrating peptides] over the past decade has been the human immunodeficiency virus type 1 (HIV-1) transcriptional activator, the TAT protein, a virally-encoded regulatory factor essential for viral replication [4]. Many different studies have now confirmed that the highly basic region located between residues 47–57 is necessary and sufficient for intracellular import and delivery of a variety of proteins and nucleic acids [3,5,6]. *In addition to the TAT peptide, numerous natural and synthetic CPPs* have been described in the literature (i.e. penetratrin [7], Pep-1/Chariot [8], and polyarginine-containing peptides [9,10,11]) and are now commercially available. *Variants on this theme* include certain cyclic polyarginine peptides with high cell permeability … [12,13,14].”
*This history of research indicates that the polyarginine motif on Spike is worth considering.
… “Cationic cell-penetrating peptides (CPPs) have been broadly used for the delivery of various types of molecular cargoes such as small molecule drugs, siRNAs, and phosphopeptides (reviewed in [1,2,29]). Most of these compounds contain a polybasic domain responsible for transport into the intracellular space. The initial, and still the best characterized CPP, is the trans-acting activator of transcription (TAT) peptide derived from the human immunodeficiency virus [30,31]. Exhaustive analyses have demonstrated that the sequence responsible for its cellular uptake consists of the arginine-rich region YGRKKRRQRRR located between residues 47 and 57 [3,29]. The relevance of the arginine residues to uptake was clearly demonstrated by the assay of truncated analogs of HIV-1 TAT47-57 [10]. The practical applications of the use of this peptide in vivo have been previously established [32]. In this latter study, Schwarze and colleagues injected a fusion protein composed of HIV-1 TAT47-57 and β-galactosidase intraperitoneally into mice, and subsequently detected significant local β-galactosidase activities in most of the tissues* analyzed.”
*AKA, stays-in-the-arm
“Aside from HIV-1 TAT47-57, a variety of other polyarginine-containing peptides have been proposed for the intracellular delivery of nucleic acids, proteins, and drugs [33,34]. … Simple arginine-rich peptides themselves have been also proposed for use as transfection reagents* since they enter cells efficiently [9,10,36,37,38].
*Pausing here to highlight what transfection reagents are:
“However, polyarginine-containing peptides are known to potently inhibit several members of the proprotein convertase (PC) family, such as furin, PC5/6, PACE4 and PC7 [ [15,16,28,39,40,41]. The results shown here strongly support the idea that the HIV-1 TAT47-57 peptide and Chariot transfection reagent do possess the off-target effect of inhibiting furin (and likely other proprotein convertases)… This off-target effect must be taken into account in in vivo therapeutic applications of polyarginine-containing CPP compounds.”
-
Select Citations:
1. Joliot A, Prochiantz A (2004) Transduction peptides: from technology to physiology. Nat Cell Biol 6: 189–196. [PubMed]
2. Verdurmen WP, Brock R (2011) Biological responses towards cationic peptides and drug carriers. Trends Pharmacol Sci 32: 116–124. 10.1016/j.tips.2010.11.005 [PubMed]
29. Foerg C, Merkle HP (2008) On the biomedical promise of cell penetrating peptides: limits versus prospects. J Pharm Sci 97: 144–162. [PubMed]
34. Tung CH, Weissleder R (2003) Arginine containing peptides as delivery vectors. Adv Drug Deliv Rev 55: 281–294. [PubMed]
9, Futaki S, Suzuki T, Ohashi W, Yagami T, Tanaka S, et al. (2001) Arginine-rich peptides. An abundant source of membrane-permeable peptides having potential as carriers for intracellular protein delivery. J Biol Chem 276: 5836–5840. [PubMed]
36. Futaki S, Ohashi W, Suzuki T, Niwa M, Tanaka S, et al. (2001) Stearylated arginine-rich peptides: a new class of transfection systems. Bioconjug Chem 12: 1005–1011. [PubMed]
38. Mitchell D, Kim D, Steinman L, Fathman C, Rothbard J (2000) Polyarginine enters cells more efficiently than other polycationic homopolymers. J Peptide Res 56: 318–325. [PubMed]
-
-
2012: Arginine-rich cell-penetrating peptides deliver gene into living human cells
Betty Revon Liu 1, Ming-Der Lin, Huey-Jenn Chiang, Han-Jung Lee. Gene. 2012 Aug 15;505(1):37-45. DOI: 10.1016/j.gene.2012.05.053
“Transgenesis is a process that introduces exogenous nucleic acids into the genome of an organism to produce desired traits or evaluate function. Improvements of transgenic technologies are always important pursuit in the last decades. Recently, cell-penetrating peptides (CPPs) were studied as shuttles that can internalize into cells directly and serve as carriers to deliver different cargoes into cells. In the present study, we evaluate whether arginine-rich CPPs can be used for gene delivery into human cells in a noncovalent fashion. We demonstrate that three arginine-rich CPPs (SR9, HR9, and PR9) are able to transport plasmid DNA into human A549 cells. … Our results suggest that arginine-rich CPPs, especially HR9, appear to be a high efficient and promising tool for gene transfer.”
-
2011: A gene delivery system for insect cells mediated by arginine-rich cell-penetrating peptides.
Chen YJ, Liu BR, Dai YH, Lee CY, Chan MH, Chen HH, Chiang HJ, Lee HJ.Gene. 2012 Feb 10;493(2):201-10. doi: 10.1016/j.gene.2011.11.060.
-
2011: Gene transport and expression by arginine-rich cell-penetrating peptides in Paramecium.
Dai YH, Liu BR, Chiang HJ, Lee HJ.Gene. 2011 Dec 10;489(2):89-97. doi: 10.1016/j.gene.2011.08.011.
-
2017: Arginine-rich membrane-permeable peptides are seriously toxic.
Li Q, Xu M, Cui Y, Huang C, Sun M.Pharmacol Res Perspect. 2017 Oct;5(5):e00334. doi: 10.1002/prp2.334.
“The membrane-permeable peptides (MPP) such as undecapeptides TAT (YGRKKRRQRRR) and CTP (YGRRARRRRRR) have been receiving much attention for delivering various kinds of low membrane-permeability materials in vitro and in vivo. We have successfully used MPP in carrying various proteins through blood-brain barrier (BBB) in treatment of many kinds of nervous diseases. However, people always concentrate their mind on the efficacy and the mechanism of permeation of the conjugates across BBB, but overlook the toxicity of the membrane-permeable peptide itself. Once we injected intravenously not very large amounts of gamma-aminobutyric acid-MPP (GABA-MPP) to the mice, to our great surprise, the mice died within seconds with seizure, whereas the GABA control mice well survived. Thus, the importance of the toxicity of MPPs and their conjugates comes into the field of our vision. The low LD50* values of arginine-rich TAT (27.244 mg kg-1 ) and CTP (21.345 mg kg-1 ) per se in mice indicate that they all fall within the range of highly toxic chemicals.”
*LD50 = the Dose at which a substance is Lethal to 50% of the subjects tested.
“Among the arginine-rich peptides, R11 (RRRRRRRRRRR), a peptide composed purely of arginine residues, has the lowest LD50 value (16.5 mg kg-1 ) and manifests the highest toxicity… in mice. The mass percentage of arginine-rich MPP in the conjugate is critically important, the mass ratio of arginine in the MPP appears a linear correlation with the toxicity.”
-
‘Acquirement’
“A distinctive feature of the SARS-CoV-2 S-protein is the Furin cleavage site acquirement, polybasic cleavage site (RRAR) at S1–S2 junction which is cleaved efficiently by furin enzyme” (Guizani et al. 2021 (doi: 10.1007/s00011-021-01487-6) referencing Coutard et al. 2020 (doi: 10.1016/j.antiviral.2020.104742).
-
Peculiarly, Strikingly, Canonical…
The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade.
Coutard B, Valle C, de Lamballerie X, Canard B, Seidah NG, Decroly E. Antiviral Res. 2020;176:104742. doi: 10.1016/j.antiviral.2020.104742. [PMC free article])
“We identified a peculiar furin-like cleavage site in the Spike protein of the 2019-nCoV, lacking in the other SARS-like CoVs…
… Strikingly, the 2019-nCoV S-protein sequence contains 12 additional nucleotides upstream of the single Arg↓ cleavage site 1 … leading to a predictively solvent-exposed PRRAR↓SV sequence, which corresponds to a canonical furin-like cleavage site (Braun and Sauter, 2019; Izaguirre, 2019; Seidah and Prat, 2012). This furin-like cleavage site is supposed to be cleaved during virus egress (Mille and Whittaker, 2014) for S-protein “priming” and may provide a gain-of-function to the 2019-nCoV for efficient spreading in the human population compared to other lineage b betacoronaviruses.”
*With interest in Zika’s peculiar and striking use of other canonical lab motifs, see my posts e.g. ‘Wrecking Ball’ or ‘Heat Shock Protein’…
-
-
A Multibasic Cleavage Site in the Spike Protein of SARS-CoV-2 Is Essential for Infection of Human Lung Cells
Markus Hoffmann, Hannah Kleine-Weber, Stefan Pöhlmann. Vol. 78:4, P779-784.E5, May 21, 2020
“The viral spike protein mediates SARS-CoV-2 entry into host cells and harbors a S1/S2 cleavage site containing *multiple arginine residues (multibasic)* not found in closely related animal coronaviruses. … Here, we report that the cellular protease furin cleaves the spike protein at the S1/S2 site and that cleavage is essential for S-protein-mediated cell-cell fusion and entry into human lung cells.
…These results indicate that the presence of several arginine residues at the S1/S2 site is required for efficient SARS-2-S proteolytic processing in human cells and also confers high cleavability to SARS-S….
Furin Cleaves the SARS-CoV-2 Spike Protein at the S1/S2 Site, and Cleavage Is Required for Efficient Cell-Cell Fusion
…deletion of the multibasic motif resulted in a spike protein that was no longer able to induce syncytium formation even in the presence of trypsin or TMPRSS2. Finally, the **addition of an arginine residue to the S1/S2 site of SARS-2-S jointly with alanine-to-lysine exchange strongly increased syncytium formation**…”
-
-
SYNCYTIA
Syncytium: A multinucleated mass, caused for instance by multiple cell fusions. (Cardiac action potential and the astrocytic blood brain barrier require functional syncytium performance to keep things running — what might go wrong if syncytia form randomly?)
-
-
Syncytia in HIV. Is Tat involved?
Necroptosis takes place in human immunodeficiency virus type-1 (HIV-1)-infected CD4+ T lymphocytes.
Pan T, Wu S, He X, et al. PLoS One. 2014;9(4):e93944. Published 2014 Apr 8. doi:10.1371/journal.pone.0093944
Human immunodeficiency virus type 1 (HIV-1) infection is characterized by progressive depletion of CD4+ T lymphocytes and dysfunction of the immune system. … In this report, we demonstrate that HIV-1 not only induces apoptosis, but also mediates necroptosis in the infected primary CD4+ T lymphocytes and CD4+ T-cell lines. Necroptosis-dependent cytopathic effects are significantly increased in HIV-1-infected Jurkat cells. … Treatment with necrostatin-1(Nec-1), a RIP1 inhibitor that specifically blocks the necroptosis pathway, potently restrains HIV-1-induced cytopathic effect and interestingly, inhibits the formation of HIV-induced syncytia in CD4+ T-cell lines. This suggests that syncytia formation is mediated, at least partially, by necroptosis-related processes. Furthermore, we also found that the HIV-1 infection-augmented tumor necrosis factor-alpha (TNF-α) plays a key role in inducing necroptosis and HIV-1 Envelope and Tat proteins function as its co-factors. …
… Syncytia formation is a major death pathway for CD4+ T-cell lines infected with HIV-1 and has long been considered as a marker of HIV-1 infection in these cell lines. …
… Interactions between HIV-1-infected and neighboring uninfected cells often lead to cell fusion and the formation of multi-nucleated syncytial cells, which have been observed both in vitro and in vivo in different organs including the brain [52]–[54]. The syncytia induced by specific virus strains in T cell lines are frequently correlated with the final phase of AIDS progression [42], [55], [56]. The molecular mechanism of syncytia formation remains elusive. … Our data show a strong correlation between syncytia formation and necroptosis. … At this point, we cannot conclude whether necroptosis is a cause or a consequence of syncytia formation. …
…We also found that Envelope protein or Tat can function as a co-factor for TNF-α to induce necroptosis.”
-
-
The Mechanism and Consequences of SARS-CoV-2 Spike-Mediated Fusion and Syncytia Formation
Review. Rajah MM, Bernier A, Buchrieser J, Schwartz O. J Mol Biol. 2022 Mar 30;434(6):167280. doi: 10.1016/j.jmb.2021.167280.
“Syncytia are formed when individual cells fuse. SARS-CoV-2 induces syncytia when the viral spike (S) protein on the surface of an infected cell interacts with receptors on neighboring cells. … SARS-CoV-2 variants of concern possess several mutations within the S protein that enhance receptor interaction, fusogenicity and antibody binding. …”
Image: “The potential consequences of syncytia formation on SARS-CoV-2 pathology. Center: Cells infected with SARS-CoV-2 express the spike protein at the surface and form large multinucleated syncytia. Top left: The process of syncytial death via apoptosis or pyroptosis can release virus to infect neighbouring cells and/or trigger an inflammatory response. Top right: Infected syncytia can lift off to contribute to viral dissemination and the overall infectious dose. Bottom left: Syncytial cells can target lymphocytes for cell-in-cell mediated death. Bottom right: Syncytia can facilitate cell-to-cell spread of the virus and shield the virus from neutralizing antibodies.”
-
-
Syncytia formation by SARS-CoV-2-infected cells
Buchrieser J, Dufloo J, Hubert M, Monel B, Planas D, Rajah MM, Planchais C, Porrot F, Guivel-Benhassine F, Van der Werf S, Casartelli N, Mouquet H, Bruel T, Schwartz O. EMBO J. 2020 Dec 1;39(23):e106267. doi: 10.15252/embj.2020106267. Erratum in: EMBO J. 2021 Feb 1;40(3):e107405. PMID: 33051876
“Severe cases of COVID-19 are associated with extensive lung damage and the presence of infected multinucleated syncytial pneumocytes. The viral and cellular mechanisms regulating the formation of these syncytia are not well understood. Here, we show that SARS-CoV-2-infected cells express the Spike protein (S) at their surface and fuse with ACE2-positive neighboring cells. Expression of S without any other viral proteins* triggers syncytia formation.”
*As in, spike alone, however that might end up in the arm or beyond
… “TMPRSS2 serine protease, which is known to enhance infectivity of cell-free virions, processes both S and ACE2 and increases syncytia formation by accelerating the fusion process. … Our results show that SARS-CoV-2 pathological effects are modulated by cellular proteins that either inhibit or facilitate syncytia formation.”
-
-
SARS-CoV-2 Alpha, Beta, and Delta variants display enhanced Spike-mediated syncytia formation.
Rajah MM, Hubert M, Bishop E, Saunders N, Robinot R, Grzelak L, Planas D, Dufloo J, Gellenoncourt S, Bongers A, Zivaljic M, Planchais C, Guivel-Benhassine F, Porrot F, Mouquet H, Chakrabarti LA, Buchrieser J, Schwartz O.EMBO J. 2021 Dec 15;40(24):e108944. doi: 10.15252/embj.2021108944.
“Here, we first assessed Alpha (B.1.1.7) and Beta (B.1.351) spread and fusion in cell cultures, compared with the ancestral D614G strain. … Alpha and Beta formed larger and more numerous syncytia. … Delta spike also triggers faster fusion relative to D614G. Thus, SARS-CoV-2 emerging variants display enhanced syncytia formation.”
-
-
LYMPHOCYTE ELIMINATION
-
Induction of apoptosis in uninfected lymphocytes by HIV-1 Tat protein
Li CJ, Friedman DJ, Wang C, Metelev V, Pardee AB (1995) Science 268: 429–431. DOI: 10.1126/science.7716549
“Infection by human immunodeficiency virus-type 1 (HIV-1) is typified by the progressive depletion of CD4 T lymphocytes and deterioration of immune function in most patients. A central unresolved issue in acquired immunodeficiency syndrome (AIDS) pathogenesis is the mechanism underlying this T cell depletion. HIV-1 Tat protein was shown to induce cell death by apoptosis in a T cell line and in cultured peripheral blood mononuclear cells from uninfected donors. ...”
-
-
This paper, featuring Wuhan bat scientist Shi, was covered by Igor Chudov’s Substack, Sars-Cov-2 kills T-Cells, just like HIV. This paper names LFA-1 receptor on lymphocytes, which responds to HIV GP120 (also on Spike). Tat / multi-arginine-motifs have various ways of crossing cell membranes (mentioned above by Ansari et al.) which are not considered.
doi:10.1038/s41392-022-00919-x
“SARS-CoV-2 induced marked lymphopenia in severe patients with COVID-19. …Here, we confirmed that SARS-CoV-2 viral antigen could be detected in patient peripheral blood cells (PBCs) or postmortem lung T cells … We next prove that SARS-CoV-2 infects T lymphocytes, preferably activated CD4 + T cells in vitro. … We show that the infection is spike-ACE2/TMPRSS2-independent … Viral antigen-positive T cells from patients [underwent] pronounced apoptosis. …
…Notably, lymphopenia was observed in 83.2% of the patients on admission, and fatal infections were associated with more severe lymphopenia over time. 6–8 Lymphocytes (particularly T cells) play a central role in the human immune system, a decrease of which would result in immune suppression and serious complications. 10 …”
-
-
Necroptosis takes place in human immunodeficiency virus type-1 (HIV-1)-infected CD4+ T lymphocytes.
Pan T, Wu S, He X, et al. PLoS One. 2014;9(4):e93944. Published 2014 Apr 8. doi:10.1371/journal.pone.0093944
“Syncytia formation is a major death pathway for CD4+ T-cell lines infected with HIV-1 and has long been considered as a marker of HIV-1 infection in these cell lines.” …
-
-
SARS-CoV-2 spike protein dictates syncytium-mediated lymphocyte elimination.
Zhang Z, Zheng Y, Niu Z, Zhang B, Wang C, Yao X, Peng H, Franca DN, Wang Y, Zhu Y, Su Y, Tang M, Jiang X, Ren H, He M, Wang Y, Gao L, Zhao P, Shi H, Chen Z, Wang X, Piacentini M, Bian X, Melino G, Liu L, Huang H, Sun Q. Cell Death Differ. 2021 Sep;28(9):2765-2777. doi: 10.1038/s41418-021-00782-3.
“The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus is highly contagious and causes lymphocytopenia, but the underlying mechanisms are poorly understood. We demonstrate here that heterotypic cell-in-cell structures with lymphocytes inside multinucleate syncytia are prevalent in the lung tissues of coronavirus disease 2019 (COVID-19) patients. These unique cellular structures are a direct result of SARS-CoV-2 infection, as the expression of the SARS-CoV-2 **spike glycoprotein is sufficient** to induce a rapid (~45.1 nm/s) membrane fusion to produce syncytium, which could readily internalize multiple lines of lymphocytes to form typical cell-in-cell structures, remarkably leading to the death of internalized cells. This membrane fusion is dictated by a **bi-arginine** motif within the polybasic S1/S2 cleavage site…
…We next examined whether the multinucleate syncytia produced by membrane fusion could internalize lymphocytes to form heterotypic cell-in-cell structures, CCRF-CEM, a human lymphoblast cell line isolated from a child with acute T cell leukemia… Syncytia with more nuclei or those that were larger in size were more likely to internalize CCRF-CEM cells … In addition to CCRF-CEM, these syncytia could also efficiently internalize multiple types of leukocytes, including THP-1 cells, a monocyte cell line from an acute monocytic leukemia patient; Jurkat cells, a T lymphocytes cell line from an acute T cell leukemia patient; Raji cells, a B lymphocyte cell line from a Burkitt’s lymphoma patient; K562 cells, a human erythroleukemia line from a chronic myelogenous leukemia patient; as well as peripheral blood mononuclear cells (PBMC) isolated from a healthy man (Fig. 3e, f). … The syncytia-mediated death turned out to be an efficient way to eliminate PBMC in a coculture experiment, where the amount of PBMC progressively decreased when cocultured with 293T-ACE2 cell expressing spike protein (293T-ACE2 spike) leading to syncytia formation; and by 24 h of coculture, more than 30% PBMC were cleared in 293T-ACE2 spike coculture as compared with those in control coculture (293T-ACE2 vector) …. The internalized PBMC were mostly positive in CD8, and CD4 to a less extent … These results are consistent with an efficient clearance of lymphocytes, preferentially CD8+ T cells, by syncytia….
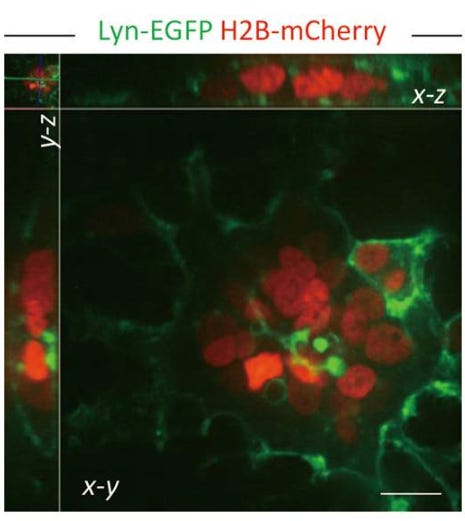
Photo: Representative images of a syncytium formed in 293T-ACE2 cells expressing exogenous SARS-CoV-2 spike glycoprotein, Lyn-EGFP (cell membrane, green), and H2B-mCherry (nucleus, red). Scale bars: 20 μm….
Image: A working model for SARS-CoV-2-induced lymphocyte loss via syncytia-mediated cell-in-cell formation.
… The infection of ACE2-expressing cells by SARS-CoV-2 virus leads to the surface expression of viral spike glycoprotein, which harbors a *bi-arginine motif* that is required for protease-mediated processing and *controls membrane fusion*. The engagement of spike protein with its receptor ACE2 triggers membrane fusion, mediated by the S2 domain of the viral spike glycoprotein, between the neighboring cells, leading to the production of multinucleated syncytium. The syncytia are capable of targeting lymphocytes for internalization and cell-in-cell mediated death, conceivably contributing to lymphopenia in COVID-19 patients.
-
-
Below Image: Massive (buffy coat color) protein clots found in autopsies. Dr. Ryan Cole, Cole Diagnostics. Featured in an article by Andrea Tice, 1819 News, July 30 2022.
-
My next post will expand on the theme of HIV-TAT / Arginine / Syncytia by discussing extracellular matrix changes. These dynamics interacting together could point to some of the pathological phenomena seen in lung / brain / etc. damage, the death processes, and autopsies [conglomerate material instead of well-defined cell ecosystem with proper electrical conductance].
-
TRYING TO BLOCK SYNCYTIA
-
Spike alone … HIV…
The anti-HIV drug nelfinavir mesylate (Viracept) is a potent inhibitor of cell fusion caused by the SARSCoV-2 spike (S) glycoprotein warranting further evaluation as an antiviral against COVID-19 infections.
Musarrat F, Chouljenko V, Dahal A, Nabi R, Chouljenko T, Jois SD, Kousoulas KG.J Med Virol. 2020 Oct;92(10):2087-2095. doi: 10.1002/jmv.25985.
“The spike (S) glycoprotein is a major determinant of virus infectivity. Herein, we show that the transient expression of the SARS CoV-2 S glycoprotein in Vero cells caused extensive cell fusion (formation of syncytia) in comparison to limited cell fusion caused by the SARS S glycoprotein.”
-
-
Evolved, passive voice
Design of Potent Membrane Fusion Inhibitors against SARS-CoV-2, an Emerging Coronavirus with High Fusogenic Activity.
Zhu Y, Yu D, Yan H, Chong H, He Y.J Virol. 2020 Jul 1;94(14):e00635-20. doi: 10.1128/JVI.00635-20.
“Herein, we report that the SARS-CoV-2 S protein has evolved a high level of activity to mediate cell-cell fusion, significantly differing from the S protein of SARS-CoV that emerged previously.”
-
Enfuvirtide, an HIV-1 fusion inhibitor peptide, can act as a potent SARS-CoV-2 fusion inhibitor: an in silico drug repurposing study.
Ahmadi K, Farasat A, Rostamian M, Johari B, Madanchi H.J Biomol Struct Dyn. 2022 Aug;40(12):5566-5576. doi: 10.1080/07391102.2021.1871958.
-
Structural insight into SARS-CoV-2 neutralizing antibodies and modulation of syncytia.
Asarnow D, Wang B, Lee WH, Hu Y, et al. Cell. 2021 Jun 10;184(12):3192-3204.e16. doi: 10.1016/j.cell.2021.04.033.
-
Computationally prioritized drugs inhibit SARS-CoV-2 infection and syncytia formation.
Serra A, Fratello M, Federico A, Ojha R, Provenzani R, et al. Brief Bioinform. 2022 Jan 17;23(1):bbab507. doi: 10.1093/bib/bbab507.
-
Anti-Fungal Drug Anidulafungin Inhibits SARS-CoV-2 Spike-Induced Syncytia Formation by Targeting ACE2-Spike Protein Interaction.
Ahamad S, Ali H, Secco I, Giacca M, Gupta D.Front Genet. 2022 Mar 25;13:866474. doi: 10.3389/fgene.2022.866474.
-
-
April 2020:
Inhibition of SARS-CoV-2 (previously 2019-nCoV) infection by a highly potent pan-coronavirus fusion inhibitor targeting its spike protein that harbors a high capacity to mediate membrane fusion
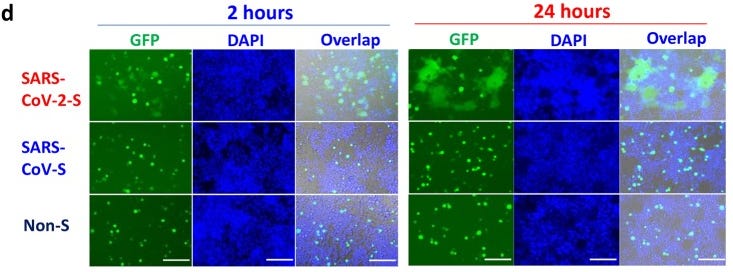
Xia S, Liu M, Wang C, Xu W, Lan Q, Feng S, Qi F, Bao L, Du L, Liu S, Qin C, Sun F, Shi Z, Zhu Y, Jiang S, Lu L. Cell Res. 2020 Apr;30(4):343-355. doi: 10.1038/s41422-020-0305-x. Epub 2020 Mar 30.
d Images of SARS-CoV and SARS-CoV-2 S-mediated cell–cell fusion on 293T/ACE2 cells at 2 h (left) and 24 h (right).
-
-
BRAIN INJURY
Above, we saw that Tat and Tat-similar Arginine motifs were equally capable of various actions [permeating membranes, delivering gene loads, triggering syncytia, etc.] We also saw that Spike has Arginine motifs and achieves some of the same actions. This section starts by showing that Spike passes the BBB, then presents a few articles about Tat-triggered brain damage — knowledge which has emerged over decades of observation.
My next post will bring in extracellular matrix disruption [Spike, Tat, MMPs etc.] to expand on brain injury and tissue-remodeling mechanisms.
-
-
We started this post with Kanazawa et al. (2013) telling how to penetrate the Blood-Brain Barrier using arginine-rich Tat and PEG. Here’s Spike protein:
The S1 protein of SARS-CoV-2 crosses the blood–brain barrier in mice.
Rhea, E.M., Logsdon, A.F., Hansen, K.M. et al. Nat Neurosci 24, 368–378 (2021). https://doi.org/10.1038/s41593-020-00771-8
“We show that intravenously injected radioiodinated S1 (I-S1) [S1 subunit of spike protein] readily crossed the blood–brain barrier in male mice, was taken up by brain regions and entered the parenchymal brain space. I-S1 was also taken up by the lung, spleen, kidney and liver.
…I-S1 from two different commercial sources readily crosses the mouse BBB, at least when injected intravenously. I-S1 was taken up by all 11 brain regions examined. Such widespread entry into brain of I-S1 could explain the diverse effects of S1 and/or SARS-CoV-2 such as encephalitis, respiratory difficulties and anosmia (1,3,4). … For transport across the BBB, viral binding proteins often behave similarly to the virus itself. *For example*, interactions (including binding and transport) between the HIV-1 glycoprotein gp120* and the BBB are similar to those for the complete virus (18,28). Additionally, many if not most viral proteins themselves can be biologically highly active; for example, gp120 is highly toxic (11,12,13,14,15,16,17). …”
*To happen to choose a random example …
Speaking tactfully, how might Spike alone end up in the body’s system?…
“… It is possible that during infection by SARS-CoV-2, shed S1 is available to cross the BBB, triggering responses in the brain itself, without necessarily involving crossing of intact virus particles.
…A crucial question which we partially answered here was: what receptor does I-S1 use to enter brain and other tissues? … The mice used here only expressed murine receptors, so our findings suggest that the assumption that ACE2 must be the human protein is incorrect…”
… Inflammation induced by LPS injection* increased the amount of intravenously injected I-S1 entering the brain… LPS-exposed mice had higher I-S1 uptake in lung but lower uptake in spleen and liver; the latter likely explains why these mice had reduced I-S1 clearance from blood. Notably, this decrease in clearance from blood observed in mice in an inflammatory state* suggests that all tissues will be exposed to higher S1 levels than in the noninflammatory state…”
*Remember that constituents as well as peptide motifs in the shot are designed to provoke inflammation with the goal of inducing immune memory.
-
-
Single… Quadruple…
Enduring cortical alterations after a single in-vivo treatment of HIV-1 Tat.
Wayman WN, Dodiya HB, Persons AL, Kashanchi F, Kordower JH, Hu XT, Napier TC.Neuroreport. 2012 Oct 3;23(14):825-9. doi: 10.1097/WNR.0b013e3283578050.
-
Syncytia, HIV, Brain
HIV and HIV dementia
Dennis L. Kolson and Francisco González-Scarano. J Clin Invest. 2000;106(1):11-13. https://doi.org/10.1172/JCI10553.
“The neuropathological changes induced by HIV-1 infection of the CNS include pallor of the myelin sheaths, proliferation or prominence of astrocytes, and the formation of multinucleated giant cells (syncytia). Microglia and brain macrophages are the main cellular targets for HIV infection in the CNS, and syncytium formation is a signature finding for this infection, since it results from interactions between the viral glycoproteins gp120 and gp41 and the principal cellular HIV-1 receptors in microglia, CD4 and CCR5. The presence of multinucleated giant cells in the CNS is the most specific finding in HIV infection”
-
HIV-1 Tat interacts with and regulates the localization and processing of amyloid precursor protein.
Kim J, Yoon JH, Kim YS.PLoS One. 2013 Nov 29;8(11):e77972. doi: 10.1371/journal.pone.0077972.
-
HIV Tat protein and amyloid-β peptide form multifibrillar structures that cause neurotoxicity.
Hategan A, Bianchet MA, Steiner J, Karnaukhova E, Masliah E, Fields A, et al.Nat Struct Mol Biol. 2017 Apr;24(4):379-386. doi: 10.1038/nsmb.3379.
-
HIV-Tat regulates macrophage gene expression in the context of neuroAIDS.
Carvallo L, Lopez L, Fajardo JE, Jaureguiberry-Bravo M, Fiser A, Berman JW. PLoS One. 2017 Jun 22;12(6):e0179882. doi: 10.1371/journal.pone.0179882.
-
HIV-1 Tat disrupts blood-brain barrier integrity and increases phagocytic perivascular macrophages and microglia in the dorsal striatum of transgenic mice.
Leibrand CR, Paris JJ, Ghandour MS, Knapp PE, Kim WK, Hauser KF, McRae M.Neurosci Lett. 2017 Feb 15;640:136-143. doi: 10.1016/j.neulet.2016.12.073.
-
Neonatal intrahippocampal injection of the HIV-1 proteins gp120 and Tat: differential effects on behavior and the relationship to stereological hippocampal measures.
Fitting S, Booze RM, Mactutus CF.Brain Res. 2008 Sep 26;1232:139-54. doi: 10.1016/j.brainres.2008.07.032.
-
HIV-1 Tat protein directly induces mitochondrial membrane permeabilization and inactivates cytochrome c oxidase.
Lecoeur H, Borgne-Sanchez A, Chaloin O, El-Khoury R, Brabant M, et al. Cell Death Dis. 2012 Mar 15;3(3):e282. doi: 10.1038/cddis.2012.21.
-
HIV-1 TAT-mediated microglial activation: role of mitochondrial dysfunction and defective mitophagy.
Thangaraj A, Periyasamy P, Liao K, Bendi VS, Callen S, Pendyala G, Buch S. Autophagy. 2018;14(9):1596-1619. doi: 10.1080/15548627.2018.1476810.
-
If one chanced to have gp120 and arginine-rich Tat simultaneously present
Human Immunodeficiency Virus Type 1 gp120 and Tat Induce Mitochondrial Fragmentation and Incomplete Mitophagy in Human Neurons.
Teodorof-Diedrich C, Spector SA. J Virol. 2018 Oct 29;92(22):e00993-18. DOI: 10.1128/JVI.00993-18
“HIV …can cause neurological dysfunction, including neurodegeneration and neurocognitive impairment. The specific autophagy responsible for removal of damaged mitochondria (mitophagy) and mitochondrial dynamics constitute neuronal mitochondrial quality control mechanisms and are impaired in neurodegenerative disorders and numerous other diseases. The release of HIV proteins gp120 and Tat from infected cells is thought to play an important role in HIV-associated neurocognitive disorders (HAND) … We report that exposure of human primary neurons (HPNs) to HIV gp120 and Tat accelerates the balance of mitochondrial dynamics toward fission (fragmented mitochondria) … Both HIV proteins were found to inhibit mitophagic flux in human primary neurons. HIV gp120 and Tat induced mitochondrial damage and altered mitochondrial dynamics by decreasing mitochondrial membrane potential (ΔΨm). … Altered mitochondrial dynamics associated with HIV infection and incomplete neuronal mitophagy may play a significant role in the development of HAND and accelerated aging associated with HIV infection … Although HIV is unable to infect neurons, **viral proteins, including gp120 and Tat, can enter neurons and can cause neuronal degeneration and neurocognitive impairment**.
-
Disrupted Mitochondria and mitophagy. — This article was highlighted by DoorlessCarp’s Substack in an entry sharing this article title:
A single-cell atlas reveals shared and distinct immune responses and metabolism during SARS-CoV-2 and HIV-1 infections.
Pan T, Cao G, Tang E, et al. Preprint. bioRxiv. 2022;2022.01.10.475725.
“We utilized single-cell transcriptomics to perform a systematic comparison of 94,442 PBMCs from 7 COVID-19 and 9 HIV-1 + patients in an integrated immune atlas … While immune cells in both cohorts show shared inflammation and disrupted mitochondrial function, COVID-19 patients exhibit stronger humoral immunity, broader IFN-I signaling, elevated Rho GTPase and mTOR pathway activities, and downregulated mitophagy.”
-
Electrophysiological change:
HIV-1 Tat-induced changes in synaptically-driven network activity adapt during prolonged exposure
Krogh KA, Green MV, Thayer SA. Curr HIV Res. 2014;12(6):406-14. doi: 10.2174/1570162x13666150121110402.
“The HIV-1 transactivator of transcription (Tat) protein is released by infected cells and contributes to the pathogenesis of HAND [HIV-associated neurocognitive disorders]… Here we … study the effects of Tat on the spontaneous synaptic activity that occurs in networked rat hippocampal neurons in culture. Tat triggered aberrant network activity that exhibited a decrease in the frequency of spontaneous action potential bursts and Ca2+ spikes with a simultaneous increase in burst duration and Ca2+ spike amplitude. … Changes in network excitability may contribute to Tat-induced neurotoxicity in vitro and seizure disorders in vivo. Adaptation of neural networks may be a neuroprotective response to the sustained presence of the neurotoxic protein Tat and could underlie the behavioral and electrophysiological changes observed in HAND…
As might be predicted for patients undergoing viral driven synaptic changes, HIV infected patients exhibit abnormal electroencephalographic (EEG) rhythms and an increased incidence of seizures [4, 5], suggesting functional changes in synaptic networks…
HIV does not infect neurons; thus, the neurotoxicity produced by HIV in the brain is indirect and results from shed HIV proteins, secreted inflammatory cytokines and released excitotoxins [6]. While all of these factors likely act together to produce synaptic damage, a significant contributor is the HIV protein Tat [7]. Elevated levels of Tat are found in the cerebrospinal fluid (CSF) of virologically controlled HIV patients on antiretroviral therapy, presumably because expression of Tat continues in the presence of antiviral drugs after HIV DNA is integrated into the host genome [8]. … Transgenic animals that express Tat in the brain exhibit synapse loss and impaired cognitive function [10, 11]. In vitro studies have shown that Tat potentiates NMDA receptor function [12–14] leading to loss of excitatory synapses [15], increased inhibitory synapses [16] and, following prolonged exposure, neuronal death [17].
Figure a: Tat reduces action potential burst frequency and increases burst duration. Representative traces show bursts of action potentials, under whole-cell current-clamp mode, during a 90 s sweep from an untreated (⚊) neuron or from a neuron treated with 50 ng/mL Tat ( ) for 4 h. Insets show individual bursts of action potentials displayed on an expanded time scale.”
-
-
BRAINSTEM
Anti-neuronal antibodies against brainstem antigens are associated with COVID-19
Lucchese G, Vogelgesang A, Boesl F, Raafat D, Holtfreter S, Bröker BM, Stufano A, Fleischmann R, Prüss H, Franke C, Flöel A. EBioMedicine. 2022 Aug 10;83:104211. doi: 10.1016/j.ebiom.2022.104211.
Background: Understanding how SARS-CoV-2 affects respiratory centres in the brainstem may help to preclude assisted ventilation for patients in intensive care setting. … We previously predicted the involvement of three epitopes within distinct brainstem proteins: disabled homolog 1 (DAB1), apoptosis-inducing-factor-1 (AIFM1), and surfeit-locus-protein-1 (SURF1). … We used microarrays to screen serum from COVID-19 patients admitted to intensive care and compared those with controls who experienced mild course of the disease.
Findings: The results confirm the occurrence of IgG and IgM antibodies against the hypothesised epitopes in COVID-19 patients. Importantly, while IgM levels were similar in both groups, *IgG levels* were significantly elevated in severely ill patients compared to controls, suggesting a pathogenic role of IgG.
*Note: IgM is an indicator of current infection, whereas IgG indicates previous exposure to the antigen. What might this say about previous Spike exposure and antibodies against brainstem? See list of effects observed in the Pf trial for various antibodies targeting nervous system.
-
Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19.
Meinhardt J, Radke J, Dittmayer C, Franz J, Thomas C, Mothes R, Laue M, Schneider J, Brünink S, Greuel S, Lehmann M, Hassan O, Aschman T, Schumann E, Chua RL, Conrad C, Eils R, Stenzel W, Windgassen M, Rößler L, Goebel HH, Gelderblom HR, Martin H, Nitsche A, Schulz-Schaeffer WJ, Hakroush S, Winkler MS, Tampe B, Scheibe F, Körtvélyessy P, Reinhold D, Siegmund B, Kühl AA, Elezkurtaj S, Horst D, Oesterhelweg L, Tsokos M, Ingold-Heppner B, Stadelmann C, Drosten C, Corman VM, Radbruch H, Heppner FL. Nat Neurosci. 2021; 24:168-175. DOI: 10.1038/s41593-020-00758-5
“… Widespread dysregulation of the cardiovascular, pulmonary and renal systems has been thought to be a leading cause of disease in severe or lethal COVID-19 cases 42. In light of previous reports of infection by SARS-CoV and other CoVs in the nervous system 43 and our observations of SARS-CoV-2 in the brainstem, which comprises the primary respiratory and cardiovascular control center, it is possible that SARS-CoV-2 infection, at least in some instances, might aggravate respiratory or cardiac insufficiency—or even cause failure—in a CNS-mediated manner 44.”
-
-
Neuropathology of patients with COVID-19 in Germany: a post-mortem case series.
Jakob Matschke, MD, Marc Lütgehetmann, MD, Christian Hagel, MD, Jan P Sperhake, Prof, Ann Sophie Schröder, MD, Carolin Edler, MD,c Herbert Mushumba, MD,c Antonia Fitzek, MD, Lena Allweiss, PhD, Maura Dandri, Prof, PhD, Matthias Dottermusch, MD, Axel Heinemann, MD, Susanne Pfefferle, MD, Marius Schwabenland, MD, Daniel Sumner Magruder, MSc, Stefan Bonn, Prof, PhD, Marco Prinz, Prof, MD, Christian Gerloff, Prof, MD, Klaus Püschel, Prof, MD, Susanne Krasemann, PhD, Martin Aepfelbacher, Prof, MD, and Markus Glatzel, Prof, MD. Lancet Neurol. 2020 Nov;19(11):919-929. doi: 10.1016/S1474-4422(20)30308-2.
“…We detected fresh territorial ischaemic lesions in six (14%) patients. 37 (86%) patients had astrogliosis in all assessed regions. Activation of microglia and infiltration by cytotoxic T lymphocytes was most pronounced in the brainstem and cerebellum, and meningeal cytotoxic T lymphocyte infiltration was seen in 34 (79%) patients. SARS-CoV-2 could be detected in the brains of 21 (53%) of 40 examined patients, with SARS-CoV-2 viral proteins found in cranial nerves originating from the lower brainstem and in isolated cells of the brainstem.”
-
-
Long COVID: current status in Japan and knowledge about its molecular background.
Matsunaga A, Tsuzuki S, Morioka S, Ohmagari N, Ishizaka Y. Glob Health Med. 2022 Apr 30;4(2):83-93. doi: 10.35772/ghm.2022.01013.
“…Matschke et al. found SARS-CoV-2 viral proteins in cranial nerves originating from the lower brainstem and in isolated cells of the brainstem (41), whereas Meinhardt et al. detected RNA and the spike protein of SARS-CoV-2 in the medulla oblongata where the primary respiratory and cardiovascular control center is located (40). SARS-CoV-2 can enter the nervous system by crossing the neural-mucosal interface in the olfactory mucosa. Immunohistochemical analysis with anti-spike protein antibodies identified positive staining in cortical neurons (28).
-
-
SUDDEN DEATH
-
The gliocentric hypothesis of the pathophysiology of the sudden infant death syndrome (SIDS)
Mitterauer, B. J. (2011). Medical Hypotheses, 76(4), 482–485. doi.org/10.1016/j.mehy.2010.11.026
“The hypothesis is based on glial–neuronal interactions in the cardio-respiratory centre of the brainstem. Recently, it has been experimentally verified that glial cells, especially astrocytes, exert a modulatory function in the maintenance of homeostasis in this brain region. In addition, astrocytes may also control the rhythms of heartbeat and breathing in a pulsatile manner. Based on a model of the glial–neuronal–vascular interactions in the networks of the cardio-respiratory centre in the brainstem, possible impairments of glial function that may be responsible for the sudden infant death syndrome (SIDS) are proposed. …
**Impairments can affect gap junctions in the astrocytic syncytium** …”
*Astrocyte nuclei targeted by Arginine-rich Tat were pictured above by Chauhan et al.
-
-
HIV-1-Tat excites cardiac parasympathetic neurons of nucleus ambiguus and triggers prolonged bradycardia in conscious rats.
Brailoiu E, Deliu E, Sporici RA, Benamar K, Brailoiu GC. Am J Physiol Regul Integr Comp Physiol. 2014;306(11):R814-R822. doi:10.1152/ajpregu.00529.2013
“The mechanisms of autonomic imbalance and subsequent cardiovascular manifestations in HIV-1-infected patients are poorly understood. We report here that HIV-1 transactivator of transcription (Tat, fragment 1-86) produced a concentration-dependent increase in cytosolic Ca(2+) in cardiac-projecting parasympathetic neurons of nucleus ambiguus* …”
*in the brain stem
“… We identified several mechanisms of the Tat-induced Ca(2+) elevation … Activation of TRPV2, nonselective cation channels, induced a **robust and prolonged neuronal membrane depolarization**, thus triggering an additional P/Q-mediated Ca(2+) entry. In vivo microinjection studies indicate a dose-dependent, prolonged bradycardic effect of Tat administration into the nucleus ambiguus of conscious rats... Our results support previous studies, indicating that Tat promotes bradycardia and, consequently, may be involved in the QT interval prolongation reported in HIV-infected patients. In the context of an overall HIV-dependent autonomic dysfunction, these Tat-mediated mechanisms may account for the higher prevalence of sudden cardiac death in HIV-1-infected patients … Our results may be particularly relevant in view of the recent findings that significant Tat levels can still be identified in the cerebrospinal fluid of HIV-infected patients with viral load suppression due to efficient antiretroviral therapy.”
-
-
Characterisation of medullary astrocytic populations in respiratory nuclei and alterations in sudden unexpected death in epilepsy
Patodia S, Paradiso B, Ellis M, et al. Epilepsy Research. Vol 157, Nov. 2019. doi.org/10.1016/j.eplepsyres.2019.106213
“Central failure of respiration during a seizure is one possible mechanism for sudden unexpected death in epilepsy (SUDEP). … Specialised glial cells identified in the medulla are considered essential for normal respiratory regulation including astrocytes with pacemaker properties in the pre-Botzinger complex and populations of subpial and perivascular astrocytes, sensitive to increased pCO2, that excite respiratory neurones.”
Astrocytes* in the VLM, functionally coupled to somatostatin-expressing pre-BötC neurones (Ikeda et al., 2017) are involved in adaptive* respiratory responses during physiological challenges* (Sheikhbahaei et al., 2018b).”
*Physiological challenges - athletes on the field? Astrocytes: see Chauhan et al. image above of Tat infecting Astrocyte nuclei.
“Altered medullary glial cell populations in sudden infant death (SIDS)* (Hunt et al., 2016) an event with many parallels to SUDEP, are hypothesised to contribute to the mechanisms of death in these cases (Mitterauer, 2011). In addition, experimental data from several SUDEP models found post-ictal **brainstem spreading depolarisation mediates the irreversible respiratory collapse** (Aiba and Noebels, 2015; Loonen et al., 2019)…”
-
-
Remember, Tat drills holes in membranes. (This article also appeared in a post by DoorlessCarp, ‘Selected research into spike protein experimental gene therapy induced cardiovascular pathology & accelerated aging (senescence)’).
SARS-CoV-2 direct cardiac damage through spike-mediated cardiomyocyte fusion
Jay Schneider, David Pease, Chanakha Navaratnarajah et al. 30 October 2020, PREPRINT (Version 1) available at Research Square [https://doi.org/10.21203/rs.3.rs-95587/v1]
“Billions of electromechanically coupled cardiomyocytes (CMs) make myocardium inherently vulnerable to pathological electromechanical short circuits caused by intercellular viral spread (4-6). Beyond respiratory illness, COVID-19 affects the heart (7) and cardiac injury and arrhythmias are serious public health concerns (8-12). By studying myocardium of a young woman who died suddenly, diagnosed postmortem with COVID-19, we discovered highly focal myocardial SARS CoV-2 infection spreading from one CM to another through intercellular junctions identified by highly concentrated sarcolemmal t-tubule viral spike glycoprotein. SARS CoV-2 permissively infected beating human induced pluripotent stem cell (hiPSC)-CMs building multinucleated cardiomyotubes (CMTs) through cell type-specific fusion driven by proteolytically-activated spike glycoprotein. Recombinant spike glycoprotein, colocalizing to sarcolemma and sarcoplasmic reticulum, produced multinucleated CMTs with pathological structure, electrophysiology and Ca2+ excitation-contraction coupling. Blocking cleavage, a peptide-based protease inhibitor neutralized SARS-CoV-2 spike glycoprotein pathogenicity. We conclude that SARS-CoV-2 spike glycoprotein, efficiently primed, activated and strategically poised during biosynthesis, can exploit the CM’s inherent membranous connectivity to drive heart damage directly, uncoupling clinically common myocardial injury from lymphocytic myocarditis, often suspected but rarely confirmed in COVID-19.
… A 35 year-old Hispanic woman, 3 months post-partum, had one week of mild fever and 68 cough, felt lightheaded, went to rest and was later found dead by her husband. … Spike glycoprotein highly concentrated in t-tubule networks of adjacent CMs suggested the possibility of cell-to-cell conduits (3). If viral spike, a membrane fusion protein, opens pores between CMs, these newly created cell-cell conduits, even if microscopic, could be precarious, functionally short circuiting electrically excitable myocardium. We formulated the following hypothesis to explain this patient’s sudden cardiac death: SARS-CoV-2, brought to the myocardium via infected immune cells, spread from one CM to another through spike glycoprotein generated conduits. Intercellular connections created by spike glycoprotein drove membrane fusion that provided the pathoanatomical substrate for aberrant electrophysiological activity, electromechanical dysfunction and fatal arrhythmia.”
-
-
Let’s conclude this post with some enthusiastic ideas
-
2015: Development of a novel AIDS vaccine: the HIV-1 transactivator of transcription [TAT] protein vaccine.
Cafaro A, Tripiciano A, Sgadari C, Bellino S, Picconi O, Longo O, Francavilla V, Buttò S, Titti F, Monini P, Ensoli F, Ensoli B.Expert Opin Biol Ther. 2015;15 Suppl 1:S13-29. doi: 10.1517/14712598.2015.1021328.
-
2009: What does the structure-function relationship of the HIV-1 Tat protein teach us about developing an AIDS vaccine?
Campbell GR, Loret EP.Retrovirology. 2009 May 25;6:50. doi: 10.1186/1742-4690-6-50.
-
HIV-1 Tat protein vaccination in mice infected with Mycobacterium tuberculosis is safe, immunogenic and reduces bacterial lung pathology.
Cafaro A, Piccaro G, Altavilla G, Gigantino V, Matarese G, Olivieri E, Ferrantelli F, Ensoli B, Palma C.BMC Infect Dis. 2016 Aug 22;16(1):442. doi: 10.1186/s12879-016-1724-7.
-
A novel recombinant protein vaccine containing the different E7 proteins of the HPV16, 18, 6, 11 E7 linked to the HIV-1 Tat (47-57) improve cytotoxic immune responses.
Mousavi T, Valadan R, Rafiei A, Abbasi A, Haghshenas MR.Biotechnol Lett. 2021 Sep;43(9):1933-1944. doi: 10.1007/s10529-021-03166-2.
-
Speaking of cytotoxic immune response…
-
-
Update, Dec 13 2022: Looks like my theory on the Syncytia and the clots / sudden death is gaining interest. Dr. McCullough and James Lyons-Weiler just put out posts on it - good work. I don’t have a paid subscription to Dr. McCullough to comment and show him my posts… The article described by Dr. McCullough was published in October, covering my theory of Cell Penetrating Peptides that I’ve been writing about this summer (see posts on HIV-Tat, Transferrin Receptor, How is a brick like a bat, etc….)
James Lyon Wyler’s reposting of Dr. McCullough’s Syncytia-clot article is not yet available on his site to link to from what I can see, so I will refer you to this other good article on his site demonstrating Spike protein in brain and heart, which is also of interest.
-
-
Charles Wright looks into the history of this technology including cell transfection + HIV-vaccine attempts. For this post’s focus, of especial interest is Mal.’s CV listing a patent for “Polyfunctional cationic cytofectins, formulations and methods for generating active cytofection: polynucleotide transfection complexes” issued 10/20/98.
-
-
The articles quoted here are purely for educational or research purposes. Please refer to the copyright of the owners. The content here is an exploratory look through medical publications - no claims are being made.