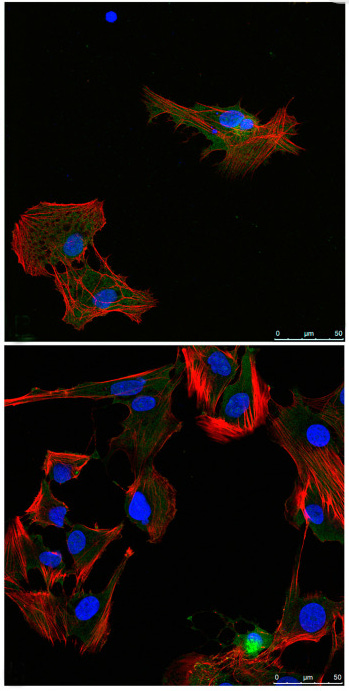
Cells melt together into Syncytia following Spike exposure. What happens to ovaries and eggs?
Spike's multi-arginine sequence, reflecting the biolab favorite gene-delivery tool HIV-1 Tat, causes syncytia.
Background:
Reviewing from the previous HIV-Tat post (linked below), Syncytia are when cells melt together. This puts a damper on basic cell functions. Action potentials in the heart and nervous system or oxygen passage in the lungs are beneficial functions that do not benefit from masses of molten cells. I am also curious whether large white clots in the vasculature have been fused as a combination of syncytia, netosis, etc.
In my previous post, I look at HIV-1 Tat protein’s multi-arginine motif as the possible source from which a similar motif in Spike was derived. This arginine-rich Spike motif has been discussed as the Furin Cleavage Site, but my interest in looking at it is from the background of HIV-Tat. Tat is long used in biolabs both as an HIV-vaccine candidate and to deliver gene transfection and bypass the body’s protective barriers.
This Post Summary:
HIV-1 Tat’s multi-arginine sequence is known to make cells melt together into Syncytia.
Braga et al. 2021: as a consequence of the multi-arginine sequence, cells with Spike on their membrane can form syncytia with other cells expressing adequate receptors.
Rajah et al. 2021: “Syncytia formation is a consequence of the S protein expressed on the surface of an infected cell *interacting with ACE2 receptors on neighboring cell.”
Wu and Ma et al. 2021: 95.58% of oocytes express ACE2
Question I haven’t seen addressed: will human eggs and new pregnancies melt into syncytia?
Likewise unaddressed: I suspect that ACE2 on decidua accounts for the decidual casts that have been observed post-vaccination. Of interest in relation to the large clots clogging vessels: sharing an underlying mechanism of Syncytia-building?
Update Dec 13, 2022: Linked at the bottom of the page are posts by Dr. McCullough and James Lyons-Weiler now talking about this theory I put out about Spike causing the giant clots via Syncytia-building.
-
Future Posts:
Commonly discussed receptors for Spike (e.g. ACE2, TMPRSS2 mentioned here) are not the only ways that Spike enters cells, therefore cell vulnerability extends beyond what is implied by focusing on these 2 proteins.
The alternatives have also been shown to be involved in Syncytia-building and are present in the reproductive organs. It would be interesting to have them all here for comparison, but the post becomes too long.
Thank you for reading.
-
-
-
Before we start, it may be helpful to visualize what it looks like when cells melt together to form syncytia:
-
Syncytia formation by SARS-CoV-2-infected cells
Buchrieser J, Dufloo J, Hubert M, Monel B, Planas D, Rajah MM, Planchais C, Porrot F, Guivel-Benhassine F, Van der Werf S, Casartelli N, Mouquet H, Bruel T, Schwartz O. EMBO J. 2021 Feb 1;40(3):e107405. doi: 10.15252/embj.2020107405.
“Here, we show that SARS-CoV-2-infected cells express the Spike protein (S) at their surface and fuse with ACE2-positive neighboring cells. Expression of S without any other viral proteins triggers syncytia formation*. … The *TMPRSS2* serine protease … processes both S and ACE2 and increases syncytia formation by accelerating the fusion process. …
*“Expression of S without any other viral proteins” / The vaccine is designed to make the body’s cells produce spike protein.
*TMPRSS2 increases / accelerates fusion - remember this below.
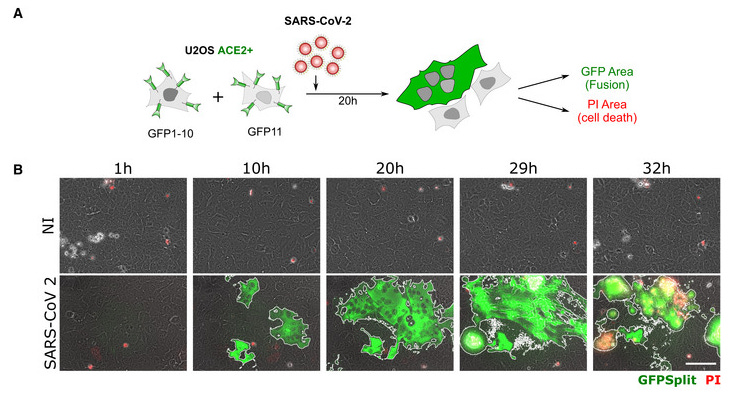
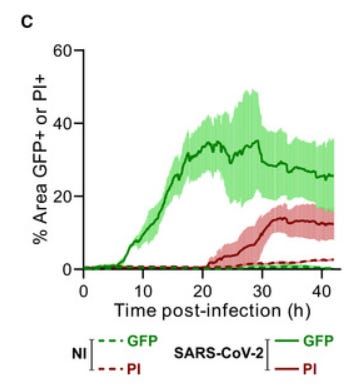
Fig. 1B: SARS‐CoV‐2 induced syncytia formation. … : Still images of GFP (syncytia, green) and propidium iodide (PI) (cell death) at different time points.
Fig. 1C [below]: Quantification of U2OS‐ACE2 fusion* (green) and death* (red) by time‐lapse microscopy. …
“Video‐microscopy analysis showed that syncytia appeared rapidly, starting at 6 h post‐infection and grew in size, as bystander cells are incorporated in fused cells ... Most of the syncytia end up dying…”
So 30 hours after Spike exposure, circa 20~45% of cells had melted together into syncytia (green), and then they started dying (red).
-
SARS-CoV-2 Alpha, Beta, and Delta variants display enhanced Spike-mediated syncytia formation
Rajah MM, Hubert M, Bishop E, Saunders N, Robinot R, Grzelak L, Planas D, Dufloo J, Gellenoncourt S, Bongers A, Zivaljic M, Planchais C, Guivel-Benhassine F, Porrot F, Mouquet H, Chakrabarti LA, Buchrieser J, Schwartz O. EMBO J. 2021 Dec 15;40(24):e108944. doi: 10.15252/embj.2021108944.
“Syncytia formation in cells expressing variant S proteins: … Syncytia formation is a consequence of the S protein expressed on the surface of an infected cell *interacting with ACE2 receptors on neighboring cell*” …
Which cells have ACE2 receptors?
-
-
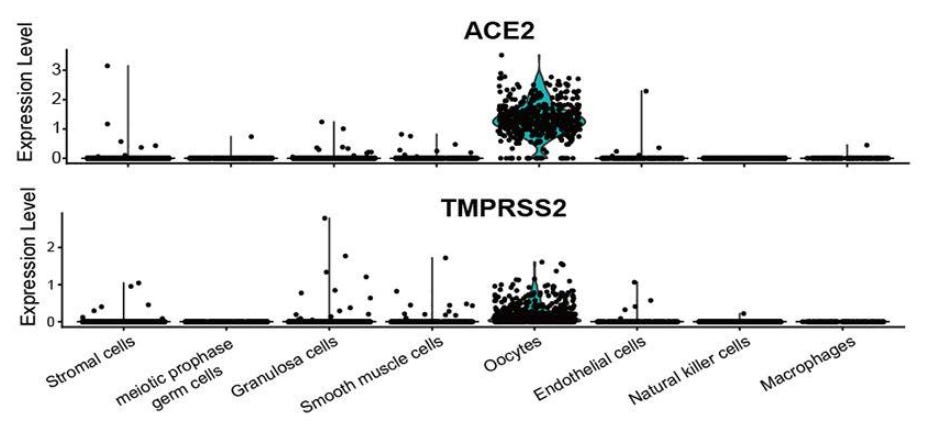
“…Predominantly enriched in oocytes…”
A closer look:
“ACE2 and TMPRSS2 were detected in the human ovarian cortex and medulla, especially in oocytes of different stages, with no observed variations in their expression level in ovaries of different ages… Remarkably, … a series of viral infection-related pathways were more enriched in ACE2-positive ovarian cells …, suggesting that SARS-CoV-2 may potentially target specific ovarian cells and affect ovarian function…
… Our study revealed that approximately 80% of the ovarian cells were positive for ACE2 and TMPRSS2; they are predominantly enriched in oocytes and matrix cells of human ovaries…
… As ovarian follicles play an essential role in ovarian function, we further explored the expression of ACE2 and TMPRSS2 in ovarian follicles of different stages. … In primordial follicles, intense ACE2 and TMPRSS2 immunostaining appeared in the oocytes. Strong immunoreactivity for ACE2 and TMPRSS2 was also observed both in oocytes and the granulosa layer of primary follicles. In secondary follicles, the granulosa cells were negative for ACE2, whereas the theca-interstitial cells showed moderate staining for ACE2, and TMPRSS2 was highly expressed in some granulosa cells. In antral follicles, ACE2 was highly expressed in the outer granulosa cells…
95.58% of oocytes expressed ACE2– the highest proportion of all cell types; 68.81% of oocytes expressed TMPRSS2.
-
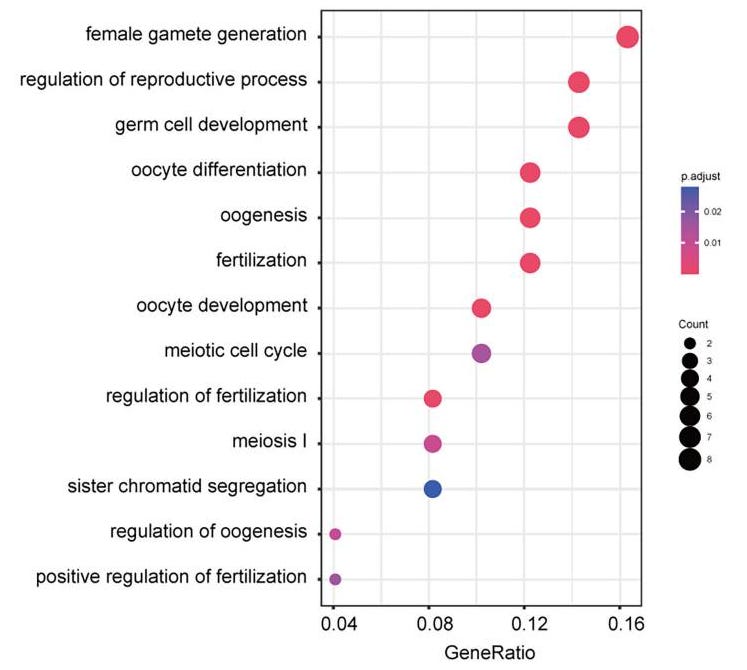
Aside from useful activities such as being the egg supply for life, what other gene processes are TMPRSS2-positive ovarian cells involved in? How might this be of interest to pregnant women who provide Spike-vaccine to their daughters?
Fig. 8 [below] “Differentially expressed gene functional analysis … in macaque single ovarian cells. Gene ontology enrichment result … of differentially expressed genes in TMPRSS2-positive and TMPRSS2-negative ovarian cells.”
-
-
-
SARS-CoV-2 Infection of Human Ovarian Cells: A Potential Negative Impact on Female Fertility
Luongo FP, Dragoni F, Boccuto A, Paccagnini E, Gentile M, Canosi T, Morgante G, Luddi A, Zazzi M, Vicenti I, Piomboni P. Cells. 2022 Apr 23;11(9):1431. doi: 10.3390/cells11091431.
“Cultured GCs [granulosa cells] and CCs [cumulus cells] incubated with SARS-CoV-2 revealed productive SARS-CoV-2 infection … TEM showed full-size virions attached to the membrane and located inside the cytoplasm. This in vitro study reveals the susceptibility of human ovarian cells to SARS-CoV-2 infection, suggesting a potential detrimental effect of COVID-19 infection on female human fertility.”
Fig. 3. Intracellular localization of SARS-CoV-2 antigens. Spike [green] in primary human granulosa cells [above] and cumulus cells [below]. …
TEM electron microscopy: … “… At 48 h post-infection, the infection became massive, and the vesicles inside the cytoplasm contained a large number of viral particles near the nuclear membrane... At this time, the first signs of cellular suffering appear and become more evident at 72 h, when the ultrastructural damage to the plasma membrane and cellular organelles, undergoing necrotic lysis as well as leakage of viral components, are clearly observed.
… We demonstrated that short-time exposures and low viral concentrations are able to cause infection. Cell damage seems to be directly related to the time of exposure; indeed, cell death is associated with prolonged exposure to SARS-CoV-2, supporting the high susceptibility of granulosa cells to viral replication. This is a very important finding that offers an opportunity to critically revise the literature related to the effects of COVID19 on female fertility. … This study provides the first biological explanation for the recently reported evidence that infection with SARS-CoV-2 could impair ovarian function, thus potentially affecting reproductive outcomes.”
-
-
Furin is upregulated by HCG administration. ACE2 is also upregulated by HCG:
Ovulatory upregulation of angiotensin-converting enzyme 2, a receptor for SARS-CoV-2, in dominant follicles of the human ovary
Choi Y, Jeon H, Brännström M, Akin JW, Curry TE Jr, Jo M. Fertil Steril. 2021 Dec;116(6):1631-1640. doi: 10.1016/j.fertnstert.2021.08.009. University of Kentucky.
Objective: To determine the temporal expression of angiotensin-converting enzyme 2 (ACE2), a receptor for SARS-CoV-2, in dominant follicles throughout the periovulatory period in women and the regulatory mechanisms underlying ACE2 expression in human granulosa/lutein cells. …
Discussion: … In women throughout their reproductive years, the ovary undergoes a series of cyclic changes during the menstrual cycle, culminating in ovulation. The preovulatory LH surge initiates these cyclic changes by inducing the expression of specific genes. The encoded products of these ovulatory genes exert their biological actions to bring about necessary cellular and extracellular changes required for ovulation and corpus luteum formation. In the present study, we showed that the expression of ACE2, …a primary receptor for SARS-CoV-2, is rapidly and dramatically induced after hCG administration in dominant follicles obtained from naturally cycling women throughout the periovulatory period. … This study provided further experimental evidence that the ovulatory induction of ACE2 expression was mediated by hCG and the hCG-induced steroid hormones progesterone and glucocorticoids in ovulatory follicles. … This novel information not only suggests the potential involvement of ACE2 as a critical enzyme for the LH surge-induced cyclic events of ovulation, cumulus expansion, oocyte maturation, and luteal formation, but also implies a possible impact of COVID-19 on vital cyclic ovarian functions and thus on women’s overall reproductive health.”
-
-
Quoting Buchreiser et al. from in vitro work above: “syncytia appeared rapidly… Most of the syncytia end up dying…” Quoting Rajah et al. again from above, “Syncytia formation is a consequence of the S protein expressed on the surface of an infected cell interacting with ACE2 receptors on neighboring cell”. Below, we see ACE2 on eggs and the fertilized pregnancy blastocysts:
-
SARS-CoV-2 host receptors ACE2 and CD147 (BSG) are present on human oocytes and blastocysts
Essahib W, Verheyen G, Tournaye H, Van de Velde H. J Assist Reprod Genet. 2020 Nov;37(11):2657-2660. doi: 10.1007/s10815-020-01952-x.
Fig. 1. “…SARS-CoV-2 host receptors ACE2 and CD147 on human oocytes and blastocysts. … CD147 (green), ACE2 (magenta) and SOX17 (grey) … 5 days post fertilization (dpf5) (g–j) and dpf6 (l–o). … Nuclei are labelled with Hoechst (blue).”
-
-
Does COVID-19 infection influence patients' performance during IVF-ET cycle?: an observational study.
Orvieto, R., Segev-Zahav, A., & Aizer, A. (2021). Gynecological Endocrinology, 37(10), 895–897. doi:10.1080/09513590.2021.1918
Orvieto et al. observed a significantly lower proportion of top-quality embryos after IVF patients recovered from COVID-19, “reflecting a detrimental effect of the COVID-19 infection on the developing gametes/embryos.”
-
-
*Another Substack writer mentioned several cases of ‘decidual cast’ occurring since the vaccine (I can’t find that quote now, let me know if you come across it). A decidual cast is when the entire womb lining sheds in one piece. How might that one piece mysteriously form?
The SARS-CoV-2 receptor, angiotensin-converting enzyme 2, is required for human endometrial stromal cell decidualization*
Chadchan SB, Popli P, Maurya VK, Kommagani R. Biol Reprod. 2021 Feb 11;104(2):336-343. doi: 10.1093/biolre/ioaa211.
“Although much of the focus is on respiratory symptoms, recent reports suggest that SARS-CoV-2 can cause pregnancy complications such as pre-term birth and miscarriages; and women with COVID-19 have had maternal vascular malperfusion and decidual arteriopathy in their placentas. Here, we report that the ACE2 protein is expressed in both endometrial epithelial and stromal cells in the proliferative phase of the menstrual cycle, and the expression increases in stromal cells in the secretory phase. … Additionally, in mice during pregnancy, the ACE2 protein was expressed in the uterine epithelial cells… Finally, progesterone induced Ace2 mRNA expression in mouse uteri … These data establish a role for ACE2 in endometrial physiology, suggesting that SARS-CoV-2 may be able to enter endometrial stromal cells and elicit pathological manifestations in women with COVID-19, including an increased risk of early pregnancy loss.”
-
-
Update, Dec 13 2022: Looks like my theory on the Syncytia and the clots / sudden death is gaining interest. Dr. McCullough and James Lyons-Weiler just put out posts on it - good work. I don’t have a paid subscription to Dr. McCullough to comment and show him my posts… The article described by Dr. McCullough was published in October, covering my theory of Cell Penetrating Peptides that I’ve been writing about this summer (see posts on HIV-Tat, Transferrin Receptor, How is a brick like a bat, etc….)
James Lyons-Weiler’s reposting of Dr. McCullough’s Syncytia-clot article is not yet available on his site to link to from what I can see, so I will refer you to this other good article on his site demonstrating Spike protein in brain and heart, which is also of interest.
-
-
-
The articles quoted here are purely for educational or research purposes. Please refer to the copyright of the owners. The content here is an exploratory look through medical publications - no claims are being made.