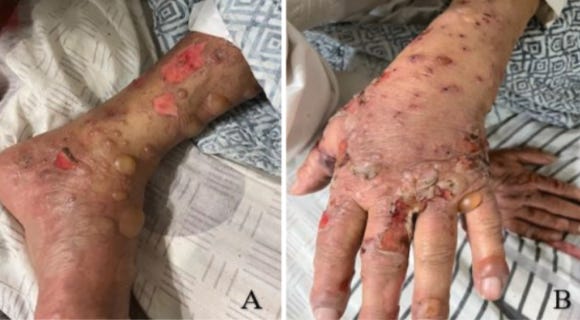
Bullous Blisters after Spike
Heat Shock Protein (HSP) epitopes on Spike, Autoimmune bullous disease. HSPs are an established target for nano-carriers.
Synopsis:
Heat Shock Protein [HSP] has long been used in bio-labs as a target to deliver e.g. nano, gene therapies, and toxic loads to cells (Elliott and Head 2012)
Spike has molecular mimicry of HSP (e.g., Lucchese and Flöel 2020)
HSP molecular mimicry may contribute to Guillain-Barré / Bullous Diseases following Spike exposure (Lucchese and Flöel 2020, Kasperkiewicz 2021)
Bullous blistering reactions have occurred after Covid vaccination (publication list below)
Antibodies against HSP have also been associated with Graft-Versus-Host-Disease, Stroke, Autoimmune vasculitis, ocular inflammation, sensorineural hearing loss, Meniere’s Disease, Myasthenia Gravis, Lupus, Rheumatoid Arthritis, Asthma, Glomerulo Nephritis, MS, Diabetes I, Schizophrenia… (publication list below)
Heat shock proteins as regulators of apoptosis and innate immunity (Joly et al. 2010); Heat shock proteins, misfolded proteins, neurodegeneration (e.g. Beretta 2022). Will be explored in a different post.)
-
2012: Heat Shock Protein as an established target to kill cells
Covered in my other post, “How is a brick like a bat?”
Below: Cancer: Tumor Iron Metabolism, Mitochondrial Dysfunction and Tumor Immunosuppression; “A Tight Partnership—Was Warburg Correct?”
Elliott RL and Head JF. 2012. DOI: 10.4236/jct.2012.34039
-
-
2016: Heat Shock Protein as an established target to kill cells
-
-
Heat-Shock Protein 90-Targeted Nano Anticancer Therapy
Ankit K Rochani 1, Aswathy Ravindran Girija 1, Ankita Borah 1, Toru Maekawa 1, D Sakthi Kumar 2
J Pharm Sci. 2016 Apr;105(4):1454-66. DOI: 10.1016/j.xphs.2015.10.007
… Nearly 2 decades of heat-shock protein 90 (Hsp90)-targeted drug discovery …. In this article, we have tried to analyze and review the Hsp90 and mammalian target of rapamycin (m-TOR)-mediated drug resistance mechanisms. … Currently, biodegradable nano vector (NV)-loaded novel drug delivery systems have shown to resolve the problems of poor bioavailability. … Looking at the success of NVs, in this article we have also discussed the progress made in the delivery of biodegradable NV-loaded Hsp90 and m-TOR-targeted inhibitors in multiple drug combinations. …
-
2020: Heat Shock Protein and Guillain-Barré
Lucchese G, Flöel A. Cell Stress Chaperones. 2020 Sep;25(5):731-735. doi: 10.1007/s12192-020-01145-6.
-
2021: Heat Shock Protein and Spike
-
Covid-19, heat shock proteins, and autoimmune bullous diseases: a potential link deserving further attention
Kasperkiewicz M. Cell Stress Chaperones. 2021 Jan;26(1):1-2. doi: 10.1007/s12192-020-01180-3.
A link between Covid-19 and development of autoimmunity has been reported. A possible explanation could be molecular mimicry between SARS-CoV-2 and human proteins. Peptide sharing has been found between antigenic epitopes of this virus and heat shock proteins (Hsp) 60 and 90, both of which are associated with autoimmune diseases including those of the bullous type. In particular, there is evidence for the latter Hsp acting as a pathophysiological factor and treatment target in autoimmune blistering dermatoses. Considering multimodal anti-inflammatory mechanisms of action of anti-Hsp90 treatment and drug repositioning results, ***it may be hypothesized that Hsp90 inhibition could also be a treatment option for cytokine storm-mediated acute respiratory distress syndrome*** in Covid-19 patients. Hence, ***although Covid-19-induced autoimmune bullous diseases have not been described in the literature so far,*** the potential relationship between Covid-19, Hsp, and these autoimmune disorders deserves further attention with respect to both pathophysiology and treatment.”
-
-
2022: HSP Inhibitors modulate Spike pathology ***
Colunga Biancatelli RML, Solopov PA, Gregory B, Khodour Y, Catravas JD.Front Physiol. 2022 Mar 21;13:812199. doi: 10.3389/fphys.2022.812199.
-
2022: Bullous diseases and Spike ***
Image: Alshammari et al. 2022: “multiple intact bullae of variable sizes with ruptured bullae and erosions.” Bullous pemphigoid after second dose of mRNA- (Pfizer-BioNTech) Covid-19 vaccine: A case report.
Alshammari F, Abuzied Y, Korairi A, Alajlan M, Alzomia M, AlSheef M.Ann Med Surg (Lond). 2022 Mar;75:103420. doi: 10.1016/j.amsu.2022.103420.
-
Can Covid‐19 vaccines cause or exacerbate bullous pemphigoid? A report of seven cases from one center.
Afacan, E., Edek, Y.C., Ilter, N., & Gülekon, A. (2022). International Journal of Dermatology, 61.
-
Akoglu G.Dermatol Ther. 2022 May;35(5):e15396. doi: 10.1111/dth.15396.
-
New‐onset bullous pemphigoid and flare of pre‐existing bullous pemphigoid after the third dose of the COVID‐19 vaccine.
Bardazzi, F., Carpanese, M.A., Abbenante, D., Filippi, F., Sacchelli, L., & Loi, C. (2022). Dermatologic Therapy, 35.
-
Calabria E, Canfora F, Mascolo M, Varricchio S, Mignogna MD, Adamo D.Pathol Res Pract. 2022 Apr;232:153834. doi: 10.1016/j.prp.2022.153834.
-
Bullous Pemphigoid Following the Moderna mRNA-1273 Vaccine.
Desai AD, Shah R, Haroon A, Wassef C.Cureus. 2022 Apr 13;14(4):e24126. doi: 10.7759/cureus.24126.
-
A case of acquired hemophilia A and bullous pemphigoid following SARS-CoV-2 mRNA vaccination.
Fu PA, Chen CW, Hsu YT, Wei KC, Lin PC, Chen TY. [published online ahead of print, 2022 Mar 14]. J Formos Med Assoc. 2022;S0929-6646(22)00098-5. doi:10.1016/j.jfma.2022.02.017
-
Gambichler T, Hamdani N, Budde H, Sieme M, Skrygan M, Scholl L, Dickel H, Behle B, Ganjuur N, Scheel C, Abu Rached N, Ocker L, Stranzenbach R, Doerler M, Pfeiffer L, Becker JC.Br J Dermatol. 2022 Apr;186(4):728-731. doi: 10.1111/bjd.20890.
-
Covid-19 dermatoses: Acral vesicular pattern evolving into bullous pemphigoid.
Goon PKC, Bello O, Adamczyk LA, Chan JYH, Sudhoff H, Banfield CC. Skin Health Dis. 2020;1(1):e6. Published 2020 Nov 11. doi:10.1002/ski2.6
-
Autoimmune Bullous Dermatosis Following COVID-19 Vaccination: A Series of Five Cases.
Hali F Sr, Araqi L Jr, Marnissi F, Meftah A, Chiheb S.Cureus. 2022 Mar 13;14(3):e23127. doi: 10.7759/cureus.23127.
-
Incident bullous pemphigoid in a psoriatic patient following mRNA-1273 SARS-CoV-2 vaccination.
Hung WK, Chi CC.J Eur Acad Dermatol Venereol. 2022 Jun;36(6):e407-e409. doi: 10.1111/jdv.17955.
-
Bullous drug eruption after second dose of mRNA-1273 (Moderna) COVID-19 vaccine: Case report.
Kong J, Cuevas-Castillo F, Nassar M, Lei CM, Idrees Z, Fix WC, Halverstam C, Mir A, Elbendary A, Mathew A.J Infect Public Health. 2021 Oct;14(10):1392-1394. doi: 10.1016/j.jiph.2021.06.021.
-
An unusual presentation of pemphigus foliaceus following COVID‐19 vaccination.
Lua, A.C., Ong, F.L., Choo, K.J., Yeo, Y.W., & Oh, C.C. (2021). Australasian Journal of Dermatology, 63.
-
Case of bullous pemphigoid following coronavirus disease 2019 vaccination.
Nakamura, K., Kosano, M., Sakai, Y., Saito, N., Takazawa, Y., Omodaka, T., Kiniwa, Y., & Okuyama, R. (2021). The Journal of Dermatology, 48.
-
Bullous pemphigoid in a young male after COVID‐19 mRNA vaccine: a report and brief literature review
M. Pauluzzi, G. Stinco, E. Errichetti. Medicine Journal of the European Academy of Dermatology and Venereology : JEADV. 2021
-
De novo severe pemphigus vulgaris following SARS-CoV-2 vaccination with BBIBP-CorV.
Saffarian Z, Samii R, Ghanadan A, Vahidnezhad H.Dermatol Ther. 2022 Jun;35(6):e15448. doi: 10.1111/dth.15448.
-
Singh A, Bharadwaj SJ, Chirayath AG, Ganguly S.J Cosmet Dermatol. 2022 Jun;21(6):2311-2314. doi: 10.1111/jocd.14945.
-
Development of severe pemphigus vulgaris following SARS-CoV-2 vaccination with BNT162b2.
Solimani F, Mansour Y, Didona D, Dilling A, Ghoreschi K, Meier K.J Eur Acad Dermatol Venereol. 2021 Oct;35(10):e649-e651. doi: 10.1111/jdv.17480.
-
Oral pemphigus after COVID‐19 vaccination.
Thongprasom, K., Pengpis, N., Phattarataratip, E., & Samaranayake, L.P. (2021). Oral Diseases.
-
A case of bullous pemphigoid after the SARS‐CoV‐2 mRNA vaccine.
Young, J., Mercieca, L., Ceci, M., Pisani, D., Betts, A., & Boffa, M.J. (2021). Journal of the European Academy of Dermatology and Venereology, 36, e13 - e16.
-
Bullous pemphigoid after inactivated COVID-19 vaccination: Case report.
Zhang, Y., Lang, X., Guo, S., He, H. and Cui, H. (2022), Dermatologic Therapy e15595. https://doi.org/10.1111/dth.15595
-
-
-
Spike, Heat Shock Protein, Bullous Disease
-
Heat Shock Protein epitopes are in Spike.
Shown above, Kasperkiewicz (Jan. 2021) draws our attention to “Peptide sharing … found between antigenic epitopes of [Sars-Cov-2] and heat shock proteins (Hsp) 60 and 90, both of which are associated with autoimmune diseases including those of the bullous type.” Below:
Mantej, Bednarek, Sitko, Świętoń, and Tukaj (Jul. 2021) checked for “autoantibodies to heat shock protein 60, 70, and 90” and found that they were “not altered” after “two doses of the mRNA vaccine” in “twenty-six healthy volunteers.”
Nevertheless, following Kasperkiewicz’s prediction, many publications report Bullous disease following Spike exposure [list above]. Would autoantibodies to heat shock protein be found in those patients?
Kasperkiewicz, Bednarek, and Tukaj (Dec. 2021) checked for “desmoglein 1, desmoglein 3, envoplakin, BP180, BP230, and type VII collagen,” the “main immunobullous autoantigens,” in a small sample size of spike-exposed people without bullous reactions and findings were negative.
However, Maronese et al. (Feb. 2022) find BP180 and BP230 antibodies in “patients with [Bullous Pemphigoid] associated with COVID-19-vaccines”.
This collection of publications demonstrates that different antibody profiles are appearing in different patients. Small sample sizes may not be sufficient to identify risks.
-
Mantej J, Bednarek M, Sitko K, Świętoń M, Tukaj S.Cell Stress Chaperones. 2021 Jul;26(4):735-740. doi: 10.1007/s12192-021-01215-3.
… It is hypothesized that the severe acute respiratory syndrome corona virus 2 (SARS-CoV-2) has the potential to elicit autoimmunity due to molecular mimicry between human extracellular Hsps and immunogenic proteins of the virus. To confirm the above hypothesis, levels of circulating autoantibodies directed to the key human chaperones i.e., Hsp60, Hsp70, and Hsp90 in the anti-SARS-CoV-2 IgG-seropositive participants have been evaluated. Twenty-six healthy volunteers who got two doses of the mRNA vaccine encoding the viral spike protein, anti-SARS-CoV-2 IgG-positive participants (n = 15), and healthy naïve (anti-SARS-CoV-2 IgG-negative) volunteers (n = 51) have been included in this study. We found that the serum levels of anti-Hsp60, anti-Hsp70, and anti-Hsp90 autoantibodies of the IgG, IgM, or IgA isotype remained unchanged in either the anti-COVID-19-immunized humans or the anti-SARS-CoV-2 IgG-positive participants when compared to healthy naïve volunteers, as measured by enzyme-linked immunosorbent assay. Our results showing that the humoral immune response to SARS-CoV-2 did not include the production of anti-SARS-CoV-2 antibodies that also recognized extracellular heat shock protein 60, 70, and 90 represent a partial evaluation of the autoimmunity hypothesis stated above. Further testing for cell-based immunity will be necessary to fully evaluate this hypothesis.
-
-
Case Report: Circulating Anti-SARS-CoV-2 Antibodies Do Not Cross-React With Pemphigus or Pemphigoid Autoantigens.
Kasperkiewicz M, Bednarek M, Tukaj S.Front Med (Lausanne). 2021 Dec 20;8:807711. doi: 10.3389/fmed.2021.807711.
“It is hypothesized that SARS-CoV-2 has the potential to elicit autoimmunity due to molecular mimicry between immunogenic proteins of the virus and human extracellular molecules. …There is limited information specifically pertaining to the immunological effects of COVID-19 and vaccines against SARS-CoV-2 on the development of autoimmune bullous diseases (AIBDs). Twelve seropositive post-COVID-19 individuals and 12 seropositive healthy volunteers who received two doses of the mRNA COVID-19 vaccine from Pfizer-BioNTech have been included in this case series investigation. Serum samples of these blood donors were tested for autoantibodies to the main immunobullous autoantigens, i.e., desmoglein 1, desmoglein 3, envoplakin, BP180, BP230, and type VII collagen. Our study revealed that none of the 24 anti-SARS-CoV-2 IgG-positive subjects had concomitant antibody reactivity with any of the tested autoantigens…These results argue against a relationship between SARS-CoV-2 infection/vaccines and AIBDs with respect to disease-triggering antibody cross-reactivity.”
-
However, other studies have found Autoimmune Bullous Diseases testing for the same antibodies:
-
Bullous Pemphigoid Associated With COVID-19 Vaccines: An Italian Multicentre Study
Maronese, C.A., Caproni, M., Moltrasio, C., Genovese, G., Vezzoli, P., Sena, P., Previtali, G., Cozzani, E., Gasparini, G., Parodi, A., Atzori, L., Antiga, E., Maglie, R., Moro, F., Mariotti, E.B., Corrà, A., Pallotta, S., Didona, B., Marzano, A.V., & di Zenzo, G. (2022). Front Med (Lausanne). 2022 Feb 28;9:841506. doi: 10.3389/fmed.2022.841506.
“Study was conducted to collect clinical, histopathological and immunopathological data of patients with BP associated with COVID-19-vaccines…. Eleven out of seventeen patients (65%) had positive titres for anti-BP180 antibodies …five out of seventeen (29%) were positive for anti-BP230 antibodies.”
-
-
If Heat Shock Protein antibodies develop, what else might be on the menu?
The frequency of HSP antibodies is high in the general population and does not always accompany manifest disease (see e.g. Kocsis et al. 2002 doi: 10.1081/imm-120016242). However, a number of studies do find elevated HSPs associated with various diseases:
-
Graft-Versus-Host Disease
Goral J, Shenoy S, Mohanakumar T, Clancy J Jr.Clin Exp Immunol. 2002 Mar;127(3):553-9. doi: 10.1046/j.1365-2249.2002.01770.x.
-
Myasthenia Gravis, Guillain-Barré
Helgeland G, Petzold A, Hoff JM, Gilhus NE, Plant GT, Romi FR.J Neuroimmunol. 2010 Aug 25;225(1-2):180-3. doi: 10.1016/j.jneuroim.2010.04.024.PMID: 20554026
-
Multiple Sclerosis
Salvetti M, Ristori G, Buttinelli C, Fiori P, Falcone M, Britton W, Adams E, Paone G, Grasso MG, Pozzilli C.J Neuroimmunol. 1996 Apr;65(2):143-53. doi: 10.1016/0165-5728(96)00013-6.PMID: 8964896
-
Stroke
Increased levels of antibodies against heat shock proteins in stroke patients.
Banecka-Majkutewicz Z, Grabowski M, Kadziński L, Papkov A, Węgrzyn A, Banecki B.Acta Biochim Pol. 2014;61(2):379-83. Epub 2014 Jun 6.PMID: 24904931
-
Acute Mania
Anti-heat shock protein 90 is increased in acute mania.
Shen WW, Liu HC, Yang YY, Lin CY, Chen KP, Yeh TS, Leu SJ.Aust N Z J Psychiatry. 2006 Aug;40(8):712-6. doi: 10.1080/j.1440-1614.2006.01872.x.PMID: 16866768
-
Schizophrenia (wait 20 years)
Kim JJ, Lee SJ, Toh KY, Lee CU, Lee C, Paik IH.Schizophr Res. 2001 Oct 1;52(1-2):127-35. doi: 10.1016/s0920-9964(00)00091-8.
-
Diabetes 1
T cells and autoantibodies to human HSP70 in type 1 diabetes in children.
Abulafia-Lapid R, Gillis D, Yosef O, Atlan H, Cohen IR.J Autoimmun. 2003 Jun;20(4):313-21. doi: 10.1016/s0896-8411(03)00038-6.
-
Placenta implications
Heat shock proteins and heat shock protein-antibody complexes in placental tissues.
Ziegert M, Witkin SS, Sziller I, Alexander H, Brylla E, Härtig W.Infect Dis Obstet Gynecol. 1999;7(4):180-5. doi: 10.1002/(SICI)1098-0997(1999)7:4<180::AID-IDOG3>3.0.CO;2-7.
-
Atopic Dermatitis
Sitko K, Bednarek M, Mantej J, Trzeciak M, Tukaj S.Cell Stress Chaperones. 2021 Nov;26(6):1001-1007. doi: 10.1007/s12192-021-01238-w.
-
Rheumatoid Arthritis
Autoantibodies to heat shock proteins 60, 70, and 90 in patients with rheumatoid arthritis.
Mantej J, Polasik K, Piotrowska E, Tukaj S.Cell Stress Chaperones. 2019 Jan;24(1):283-287. doi: 10.1007/s12192-018-0951-9.
-
Juvenile (Ideopathic) Arthritis
Zlacka D, Vavrincova P, Hien Nguyen TT, Hromadnikova I.J Autoimmun. 2006 Sep;27(2):81-8. doi: 10.1016/j.jaut.2006.06.006. Epub 2006 Aug 24.PMID: 16934956
-
SLE
Kenderov A, Minkova V, Mihailova D, Giltiay N, Kyurkchiev S, Kehayov I, Kazatchkine M, Kaveri S, Pashov A.Clin Exp Immunol. 2002 Jul;129(1):169-76. doi: 10.1046/j.1365-2249.2002.01887.x.PMID: 12100037
-
Asthma
Yang M, Wu T, Cheng L, Wang F, Wei Q, Tanguay RM.Respir Res. 2005 Feb 14;6(1):18. doi: 10.1186/1465-9921-6-18.PMID: 15710045
-
Glomerulonephritis
Chebotareva N, Bobkova I, Lysenko L, Neprinzeva N, Vinogradov A, Moiseev S.Cell Stress Chaperones. 2018 Nov;23(6):1229-1235. doi: 10.1007/s12192-018-0928-8.
-
Sudden sensorineural hearing loss
Prevalence and clinical significance of anticardiolipin, anti-beta2-glycoprotein-1, and anti-heat shock protein-70 autoantibodies in sudden sensorineural hearing loss.Gross M, Eliashar R, Ben-Yaakov A, Ulmansky R, Elidan J.Audiol Neurootol. 2008;13(4):231-8. doi: 10.1159/000115432. Epub 2008 Feb 7.PMID: 18259075
-
Autoimmune vasculitis, ocular inflammation, sensorineural hearing loss
Cogan's syndrome: anti-Hsp70 antibodies are a serological marker in the typical form.Bonaguri C, Orsoni J, Russo A, Rubino P, Bacciu S, Lippi G, Melegari A, Zavota L, Ghirardini S, Mora P.Isr Med Assoc J. 2014 May;16(5):285-8.PMID: 24979832
-
Meniere’s Disease
Shin SO, Billings PB, Keithley EM, Harris JP.Laryngoscope. 1997 Feb;107(2):222-7. doi: 10.1097/00005537-199702000-00015.PMID: 9023247
-
-
… HSP, Apoptosis and Innate Immunity (to explore another day) …
-
Dual role of heat shock proteins as regulators of apoptosis and innate immunity.
Joly AL, Wettstein G, Mignot G, Ghiringhelli F, Garrido C.J Innate Immun. 2010;2(3):238-47. doi: 10.1159/000296508. Epub 2010 Mar 16.PMID: 20375559 Review.
-
Articles cited here are for non-commercial education or research only and may be copyrighted