Adenovirus vectors
A short random perusal through publications on adeno viruses for gene transfer, culminating in ~puzzling mystery. Part 1
Copy-pasting from publications worth mentioning.
Keep in mind when reading that intravascular is not identical to intramuscular delivery, and adenovirus type + modifications made to it + previous immune background of the person being exposed will have different outcomes.
2021: Adenoviral vector vaccine platforms in the SARS-CoV-2 pandemic.
(Mendonça SA et al., doi.org/10.1038/s41541-021-00356-x)
“Adenoviral vector vaccines “represent some of the lead candidates currently supported by Operation Warp Speed and other government agencies for rapid translational development … The urgency of the COVID-19 pandemic has resulted in a flurry of development in viral vector and mRNA vaccines. … This has resulted in viral vector-based and mRNA vaccines obtaining emergency use authorizations more rapidly than any other vaccine technology, with seven out of the fourteen vaccines currently approved for at least emergency or limited use globally being adenoviral vector or mRNA based (8). In this review, we describe the biological features of adenoviral (Ad) vectors that position them as potential frontrunners in contrast with other technologies…”
Not significantly; not reported:
“Some viral vectors such as those based on lentiviruses are known to integrate into the host genome, potentially resulting in genotoxicity (31). … In contrast, Ads have not been reported to significantly integrate into the host genome, with the viral backbone remaining episomal (27). …”
Broad tissue tropism; high immunogenicity:
“The broad tissue tropism of Ads and their ability to drive strong expression of the target antigen helps position them as one of the most immunogenic viral vectors (35,36,37).”
-
2002: Adenovirus as an integrating vector
Mitani K and Kubo S, Review. DOI: 10.2174/1566523024605591
“Recombinant adenoviral vectors have served as one of the most efficient gene delivery vehicles in vivo thus far. … However, since adenovirus lacks native integration machinery, the goal of gene therapy obtaining permanent expression cannot be realized with current adenoviral vector systems. Recent studies have shown that replication-incompetent adenoviral vectors randomly integrate into host chromosomes at frequencies of 0.001-1% of infected cells. To improve the integration frequencies of adenoviral vectors, a variety of hybrid vectors combining the highly efficient DNA delivery of adenovirus with the integrating machinery of retroviruses, adeno-associated viruses, and transposons, have been emerging. …Furthermore, adenoviral vectors have shown potential as gene targeting vectors. These developments should eventually lead to more effective gene therapy vectors that can transduce a myriad of cell types stably in vivo.”
-
2021: Reverse-transcribed SARS-CoV-2 RNA can integrate into the genome of cultured human cells and can be expressed in patient-derived tissues
Zhang L. et al. DOI: 10.1073/pnas.2105968118
“…We investigated the possibility that SARS-CoV-2 RNAs can be reverse-transcribed and integrated into the DNA of human cells in culture … In support of this hypothesis, we found that DNA copies of SARS-CoV-2 sequences can be integrated into the genome of infected human cells. We found target site duplications flanking the viral sequences and consensus LINE1 endonuclease recognition sequences at the integration sites, consistent with a LINE1 retrotransposon-mediated, target-primed reverse transcription and retroposition mechanism. We also found, in some patient-derived tissues, evidence suggesting that a large fraction of the viral sequences is transcribed from integrated DNA copies of viral sequences, generating viral-host chimeric transcripts.”
-
2016: Viral Vectors for Gene Therapy: Current State and Clinical Perspectives
(Lukashev AN and Zamyatnin AA. DOI: 10.1134/S0006297916070063)
“One of the major obstacles in the development of gene therapy is the delivery of the effector to and into the target cell. … Viral vectors are one of the most promising methods for gene therapy delivery. Millions of years of evolution of viruses have resulted in the development of various molecular mechanisms for entry into cells, longterm survival within cells, and activation, inhibition, or modification of the host defense mechanisms at all levels.”
“… Recombinant vaccines can also be regarded as gene therapy, because the way they are developed and produced is closer to GT approaches rather than to the development of classical vaccines.” …
“Originally, adenoviruses were planned to be used for a broad spectrum of clinical tasks, from therapy to regenerative medicine [10]. Later, it turned out that even genetically inert adenoviral particles have extremely immunogenic capsids. Systemic application of adenoviral vectors is complicated by the fact that many viral components bind nonspecifically to blood components (proteins of the coagulation cascade, complement proteins, erythrocytes, platelets), leading to inactivation of the virus [11, 12]. Moreover, systemic administration of high doses of adenovirus can lead to systemic inflammatory response, which can be lethal in some extreme cases [13]…. Adenoviral vectors are now less used in gene therapy because of side effects, complexity of systematic administration, large genome size, and relative complexity of design.
-
[Above] Many viral components of adenoviruses bind nonspecifically to blood components, including proteins of the coagulation cascade - such as FX and FVII [below].
-
2020: The superior role of coagulation factor FX over FVII in adenoviral-mediated innate immune induction of the hepatocyte: an in vitro experiment.
(Ghahestani SF et al., doi: 10.5114/ceh.2020.99512)
Aim of the study: To better understanding the contribution of coagulation factors to the extent of adenovirus-mediated innate toxicity on the hepatocyte.
… Adenovirus-36 (AD) and adenovector type 5-GFP (Ad5-GFP) … were loaded with coagulation factors VII or X. … After adding AD-VII and AD-X complexes, the expression levels of innate inflammatory genes including protein kinase R (PKR), interleukin (IL)-1β, IL-8 and IL-18 were measured by Real-time PCR on a hepatocellular carcinoma cell line, HepG2.
The loading of coagulation factors VII and X on Ad5-GFP enhanced the transduction rate up to 50% and 60%, respectively, compared to the adenovector alone (30%). ... PKR expression increased significantly after treatment with all adenoviruses. … AD had a significant inflammatory impact on Hep-G2. … The expression of IL-1β, IL-8, and IL-18 were also enhanced following FX loading. …
Adenovirus loaded with FX exhibited more innate toxicity on the hepatocytes…
-
Expanding on the article above:
2021: The Inflammatory and Fibrotic Patterns of Hepatic Stellate Cells Following Coagulation Factors (VII or X)-Shielded Adenovirus Infection.
(Shiri A. et al., doi: 10.1007/s00284-020-02297-5.)
-
2014: The importance of coagulation factors binding to adenovirus: historical perspectives and implications for gene delivery
(Lopez-Gordo E. et al., Review. doi: 10.1517/17425247.2014.938637)
The interaction of human adenovirus (HAdV) serotype 5 (HAdV-5) with the blood coagulation factor X (FX) results in a high liver transduction after AdV intravascular administration, causing toxicity and limiting AdV delivery to the target tissue. However, FX also protects adenoviral vectors from neutralization by the complement system and natural antibodies, potentially benefiting adenoviral gene delivery, as neutralization results in the reduction of HAdV-5 binding to host cells.
… Better understanding of the interactions that take place in the bloodstream is essential to generate safe and efficient adenoviral vectors.
… After intravascular delivery, the reticuloendothelial system in the liver composed of KCs [Kupffer cells] acts as a barrier to remove AdVs from the bloodstream. These immune cells are able to take up low doses of HAdVs preventing them from transducing other cell types. In contrast, high doses of HAdVs saturate KCs and high levels of hepatocyte transduction can be achieved [124]. It was shown that upon HAdV uptake, KCs start to die by necrosis 10 min after intravenous HAdV administration and that they gradually disappear from the liver in the first hours after HAdV injection, leaving the liver free of KCs and ready to be transduced by the remaining HAdVs [147]. …
… Upon HAdV administration, C1q protein binds to HAdV neutralized by IgM antibodies activating the classical complement pathway and leading to the production of C3 convertase that cleaves C3 protein generating the anaphylactic C3a and opsonic C3b [157]. …Moreover, as diverse cell types such as erythrocytes or neutrophils have been shown to express complement receptors or antibody constant fraction (Fc) receptors [163,164], HAdV neutralization by IgM and opsonization by complement proteins may lead to transduction of alternative cell types and therefore a change in its biodistribution.
… Adenoviral vectors are currently very popular as gene transfer vehicles for applications in gene therapy, and in particular serotype 5 has been the most widely used HAdV. However, the discovery that HAdV-5 substantially transduces the liver after intravascular administration [33] causing an acute liver toxicity and activation of immune responses [34-36] represented a halt in the development of such vectors for transgene delivery to specific target organs that require delivery to the bloodstream or with certain exposure to blood…
-
2021: Adenovirus vector-attributed hepatotoxicity blocks clinical application in gene therapy
Wang Z and Zhang X, Review. doi.org/10.1016/j.jcyt.2021.07.013
“… Inherent liver tropism enables [Adenovirus vectors] AdVs to specifically deliver drugs/genes to the liver; however, AdVs are closely associated with acute hepatotoxicity in immunocompromised individuals, and the side effects of AdVs, which stimulate a strong inflammatory reaction in the liver and cause acute hepatotoxicity, have largely limited clinical application. Therefore, this review systematically elucidates the intimate relationship between AdVs and hepatotoxicity in terms of virus and host and precisely illustrates the accumulated understanding in this field over the past decades. This review demonstrates the liver tropism of AdVs and molecular mechanism of AdV-induced hepatotoxicity…”
-
“Regulatory”
2007: An inventory of shedding data from clinical gene therapy trials
(Schenk-Braat EA et al., doi: 10.1002/jgm.1096.)
Viruses are the most commonly used vectors for clinical gene therapy. The risk of dissemination of a viral vector into the environment via excreta from the treated patient, a phenomenon called shedding, is a major safety concern for the environment. Despite the significant number of clinical gene therapy trials that have been conducted worldwide, there is currently no overview of actual shedding data available. In this article, an inventory of shedding data obtained from a total of 100 publications on clinical gene therapy trials using retroviral, adenoviral, adeno-associated viral and pox viral vectors is presented. In addition, the experimental set-up for shedding analysis including the assays used and biological materials tested is summarized. The collected data based on the analysis of 1619 patients in total demonstrate that shedding of these vectors occurs in practice, mainly determined by the type of vector and the route of vector administration. Due to the use of non-quantitative assays, the lack of information on assay sensitivity in most publications, and the fact that assay sensitivity is expressed in various ways, general conclusions cannot be made as to the level of vector shedding. The evaluation of the potential impact and consequences of the observations is complicated by the high degree of variety in the experimental design of shedding analysis between trials. This inventory can be supportive to clinical gene therapy investigators for the establishment of an evidence-based risk assessment to be included in a clinical protocol application, as well as to national regulatory authorities for the ongoing development of regulatory guidelines regarding gene therapy.
-
-
2003: Development of an adenovirus-shedding assay for the detection of adenoviral vector-based vaccine and gene therapy products in clinical specimens.
(Wang F. et al., doi: 10.1089/10430340360464688.)
Adenoviral vectors are used widely as gene therapy and vaccine delivery systems. An adenovirus-shedding assay may be performed in clinical trials to monitor the safety of the vector and to investigate the potential relation between clinical symptoms and shed vector virus. This report describes the development and statistical performance of the shedding assay. Live adenovirus was recovered from throat swab and urine samples spiked with E1-deleted adenovirus type 5 vector expressing HIV-1 gag [Ad5HIVgag], in the presence or absence of wild-type adenovirus (WT Ad5). Samples were cultured in 293 and A549 cells, and the DNA extracted from virus culture was tested by polymerase chain reaction (PCR) for sequence identity. The results showed that the frequency of Ad5HIVgag infectivity in 293 cells by cytopathic effect (CPE) or an immunofluorescence assay (IFA) was concentration-dependent (53% for 10(2), 94% for 10(4), and 100% for 10(6) viral particles). WT Ad5 virus did not interfere with Ad5HIVgag. PCR amplisets could specifically amplify target sequences in the background of nonspecific DNA matrices and could distinguish Ad5HIVgag from wild-type adenoviruses. This assay may be used for clinical trials using adenovirus vectors as vehicles for vaccines.
-
-
Puzzling and mysterious
-
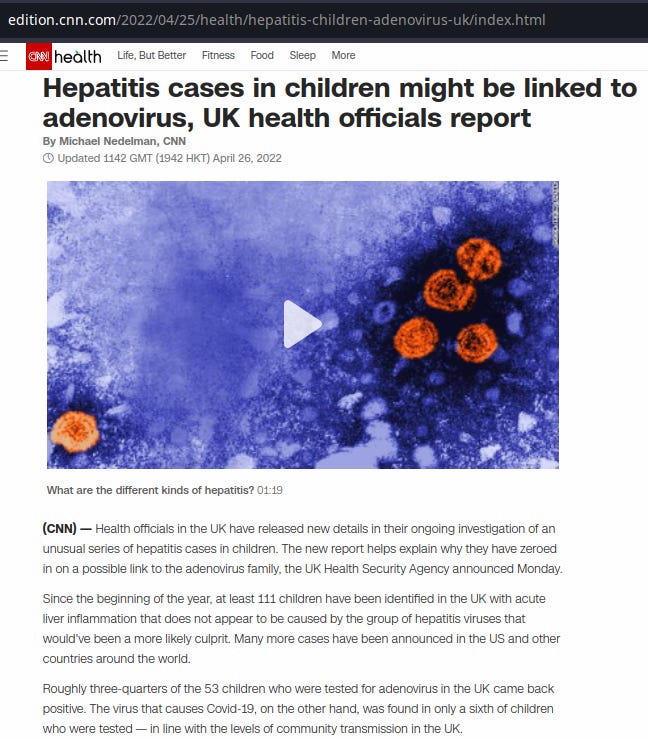
CNN, April 26, 2022, M. Nedelman:
Cofactor / coinfection / toxin / novel strain / altered characteristics / worldwide emergence:
"There may be a cofactor causing a normal adenovirus to produce a more severe clinical presentation in young children," the UK health agency said in its technical briefing Monday, "such as increased susceptibility due to reduced exposure during the pandemic, prior SARS-CoV-2 or other infection, or a yet undiscovered coinfection or toxin. Alternatively, there may have been emergence of a novel adenovirus strain with altered characteristics.”
Future topic will look at the brain implications
1995: Adenovirus gene transfer causes inflammation in the brain
(Byrnes AP et al., doi.org/10.1016/0306-4522(95)00068-T)